Physics Notes Form 4
Physics Form Four Notes
Chapter One
Thin Lences
A lens is conventionally defined as a piece of glass which is used to focus or change the direction of a beam of light passing through it.
They are mainly made of glass or plastic. Lens are used in making spectacles, cameras, cinema projectors, microscopes and telescopes.
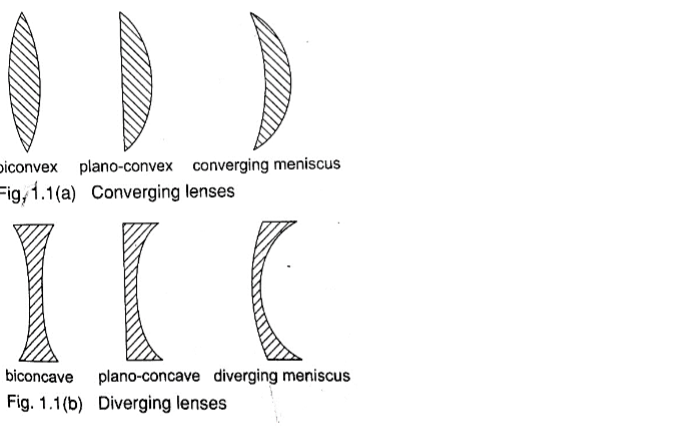
Types of thin lenses.
A lens which is thicker at its centre than at its edges converges light and is called convex or converging lens.
A lens which is thicker at its edges than at its centre diverges light and is known as concave or diverging lens.
1. Optical centre – this is the geometric centre of a lens which is usually shown using a black dot in ray diagrams. A ray travelling through the optical centre passes through in a straight line.
2. Centre of curvature – this is the geometric centre of the circle of which the lens surface is part of. Since lenses have two surfaces there are two centres of curvature. C is used to denote one centre while the other is denoted by C1.
3. Principal axis – this is an imaginary line which passes through the optical centre at right angle to the lens.
4. Principal focus – this is a point through which all rays travelling parallel to the principal axis pass after refraction through the lens.
A lens has a principal focus on both its sides. F is used to denote the principal focus
5. Focal length – this is the distance between the optical centre and the principal focus. It is denoted by ‘f’.
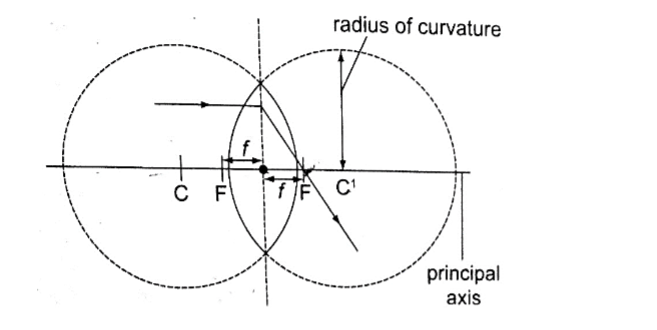
It is important to note that the principal focus is not always halfway between the optical centre and the centre of curvature as it is in mirrors.
The nature, size and position of the image formed by a particular lens depends on the position of the object in relation to the lens.
Construction of ray diagrams
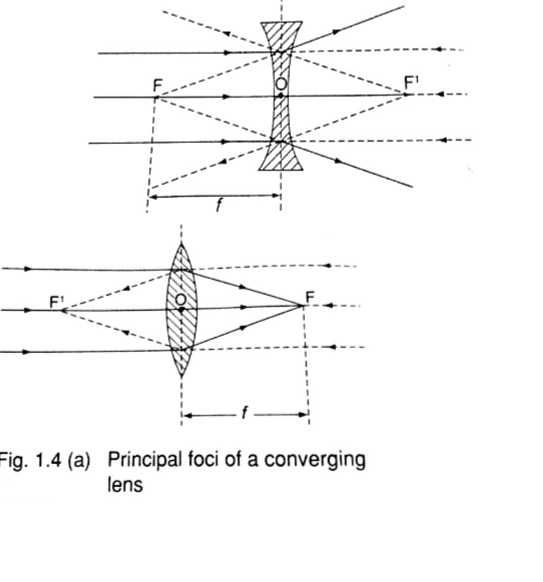
Three rays are of particular importance in the construction of ray diagrams.
1. A ray of light travelling parallel to the principal axis passes through the principal focus on refraction through the lens. In case of a concave lens the ray is diverged in a way that it appears to come from the principal focus.
2. A ray of light travelling through the optical centre goes un-deviated along the same path.
3. A ray of light travelling through the principal focus is refracted parallel to the principal axis on passing through the lens. The construction of the rays is illustrated below.
1. Object between the lens and the principal focus.
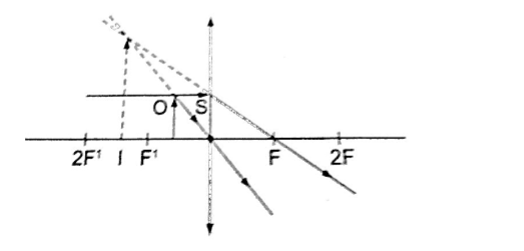
- Virtual
- Erect
- Magnified
2. Object at infinity.
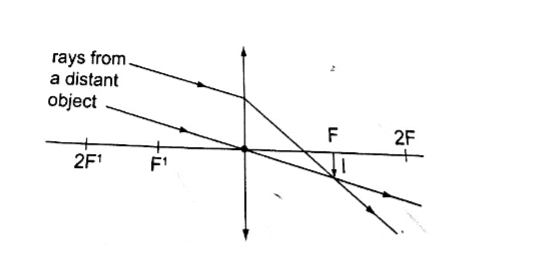
- Real
- Inverted
- Diminished
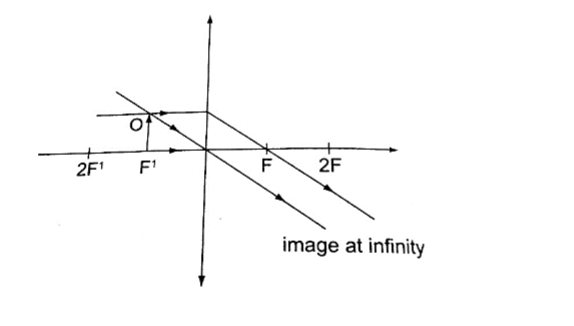
3. Object at the principal focus (at F).
4. Object between the principal focus (F) and 2 F.
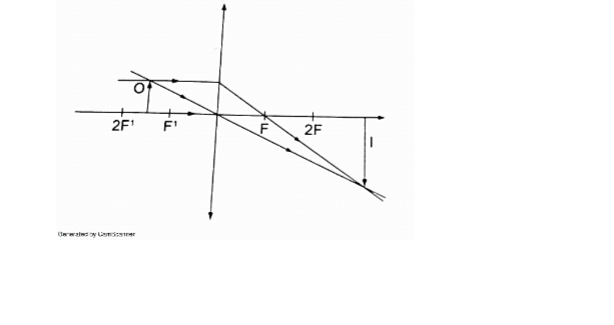
- Real
- Inverted
- Magnified
5. Object at 2 F.
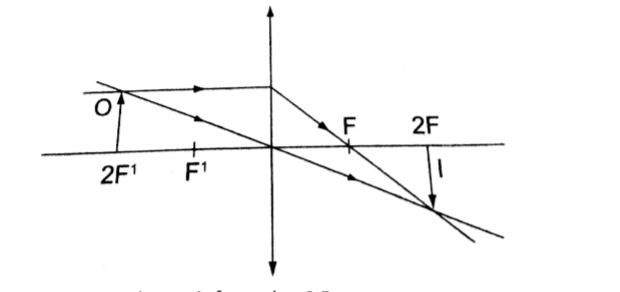
- Real
- Inverted
- Same size as the object
6. Object beyond F.
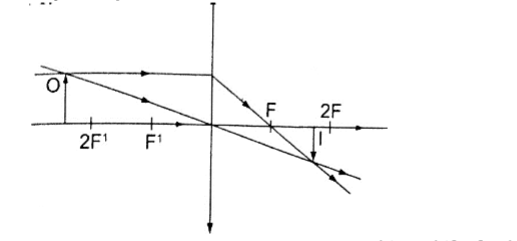
- Real
- Inverted
- Diminished
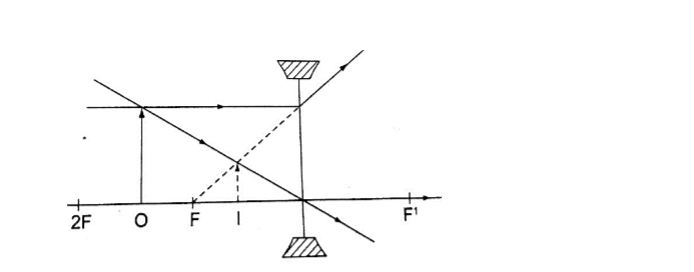
Images formed by a diverging lens.
Images formed by diverging lens are always erect, virtual and diminished for all positions of the object.
The linear magnification produced by a lens defined as the ratio of the height of the image to the height of the object, denoted by letter ‘m’, therefore;
m = height of the image / height of the object.
Magnification is also given by = distance of the image from the lens/ dist. of object from lens. m = v / u
Example
An object 0.05 m high is placed 0.15 m in front of a convex lens of focal length 0.1 m.
Find by construction, the position, nature and size of the image. What is the magnification?
Solution
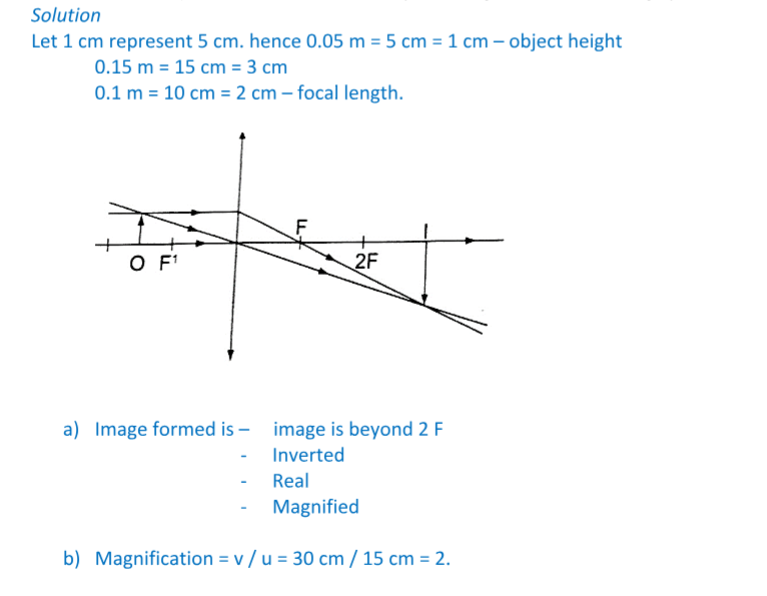
Let the object distance be represented by ‘u’, the image distance by ‘v’ and the focal length by ‘f’, then the general formula relating the three quantities is given by;
1 / f = 1 / u + 1 / v – this is the lens formula.
Examples
1. An object is placed 12 cm from a converging lens of focal length 18 cm. Find the position of the image.
Solution
Since it is a converging lens f = +18 cm (real-is-positive and virtual-is-negative rule)
The object is real therefore u = +12 cm, substituting in the lens formula, then
1 / f = 1 / u + 1 / v or 1 / v = 1 / f – 1 / u = 1 / 18 – 1 / 12 = - 1 / 36
Hence v = - 36 then the image is virtual, erect and same size as the object.
2. The focal length of a converging lens is found to be 10 cm. How far should the lens be placed from an illuminated object to obtain an image which is magnified five times on the screen?
Solution
f = + 10 cm m = v / u = 5 hence v = 5 u
Using the lens formula 1 / f = 1 / u + 1 / v => 1 /10 = 1 / u + 1 / 5 u (replacing v with 5 u)
1 / 10 = 6 / 5 u, hence 5 u = 60 giving u = 12 cm (the lens should be placed 12 cm from the illuminated object)
3. The lens of a slide projector focuses on an image of height 1.5m on a screen placed 9.0 m from the projector.
If the height of the picture on the slide was 6.5 cm, determine, a) Distance from the slide (picture) to the lens
b) Focal length of the lens
Solution
Magnification = height of the image / height of the object = v / u = 150 / 6.5 = 900 / u
u = 39 cm (distance from slide to the lens). m = 23.09
1 / f = 1 / u + 1 / v = 1 /39 + 1 / 90 = 0.02564 + 0.00111
1 / f = 0.02675 (reciprocal tables)
f = 37.4 cm.
Determining focal lengths.
1. Determining focal length of a converging lens
Experiment: To determine the focal length of a converging lens using the lens formula.
Procedure.
1. Set up the apparatus as shown below
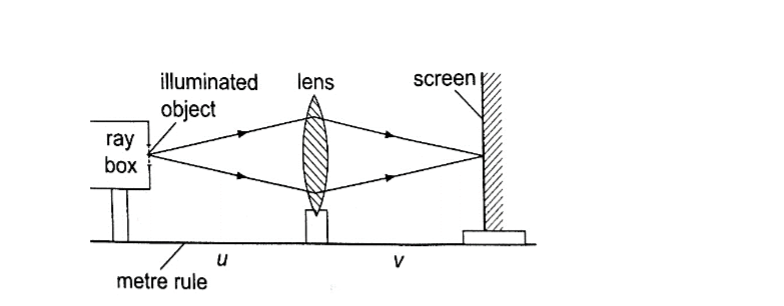
3. By changing the position of the object obtain several pairs of value of u and v and record your results as shown.

The value u v / u + v is the focal length of the lens and the different sets of values give the average value of ‘f’.
Alternatively the value ‘f’ may be obtained by plotting a graph of 1 / v against 1 / u. When plotted the following graph is obtained.
Similarly at the x-intercept, 1 / v = 0, therefore 1 / f = 1 / u or f = u hence the focal length can also be obtained by reading off the x-intercept and finding the reciprocal.
Uses of lenses on optical devices
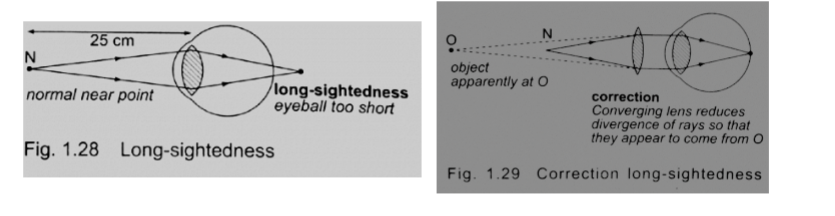
1. Simple microscope – it is also referred to as magnifying glass where the image appears clearest at about 25 cm from the eye.
This distance is known as the least distance of distinct vision (D) or near vision.
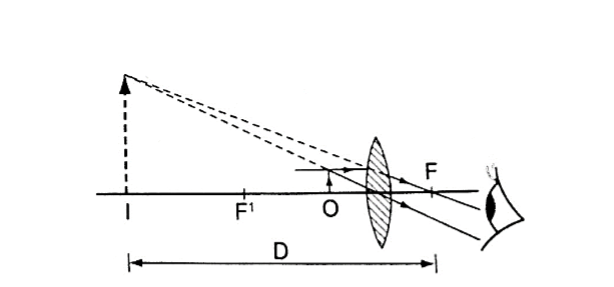
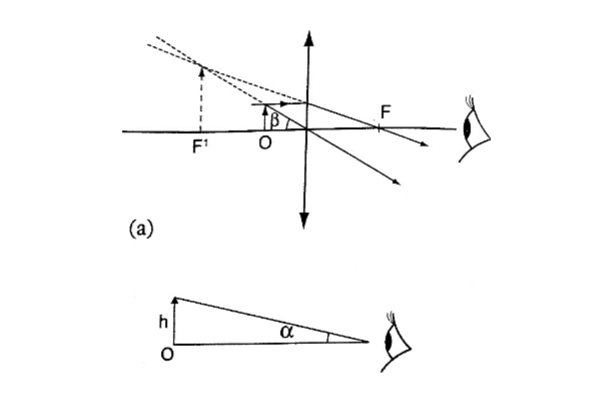
Magnification produced depends on the focal length of the lens. Lens of short focal give greater magnification than those of long focal length.
The angle ϐ subtended by the image at the eye is much greater than α which is the angle that the object would subtend at the eye when viewed without the lens.
The ratio of the ϐ toα is known as angular magnification or magnifying power of an instrument. The angular magnification is equal to linear magnification.
1. To study the features of small animals in biology
2. To look closely at small print on a map
3. To observe crystals in physics and chemistry
4. For forensic investigation by the police
2. Compound microscope - It consists of two lenses with one nearer the object called the objective lens and the other nearer the eye called the eyepiece lens.
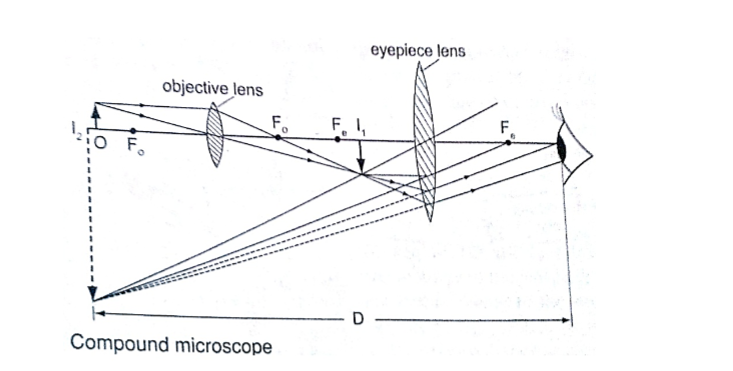
1. Used to observe Brownian motion in science
2. To study micro-organisms and cells in biology
3. Analyze laboratory tests in hospital.
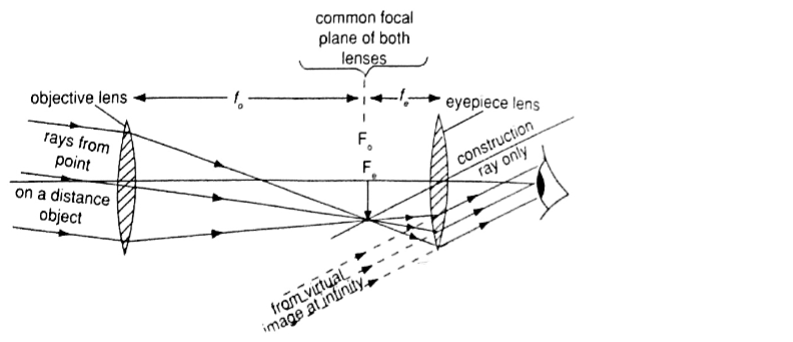
4. The astronomical telescope –It is used to view distant stars. It consists of two lenses; objective and eye-piece lenses. The objective lens has a large focal length while the eye-piece lens has a much shorter focal length.
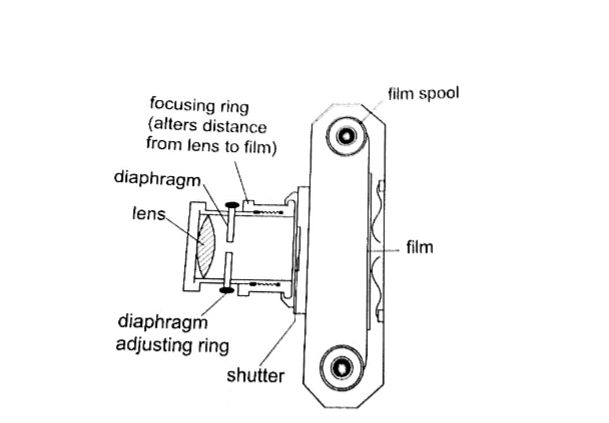
The distance is adjusted to obtain a clear focus.
The diaphragm has a hole called the aperture with an adjusting control knob to control the amount of light entering the camera. The shutter opens to allow light and close at a given time interval.
1. The sine camera is used to make motion pictures
2. High speed cameras are used to record movement of particles
3. Close circuit television cameras (CCTV) are used to protect high security installations like banks, supermarkets etc.
4. Digital cameras are used to capture data that can be fed to computers.
5. Human eye – It consists of a transparent cornea, aqueous humour and a crystal-like lens which form a converging lens system. The ciliary muscles contract or relax to change the curvature of the lens.
Though the image formed at the retina is inverted the brain ‘sees’ the image as upright.
For distant objects ciliary muscles relax while near objects it contracts to control the focal length and this is known as accommodation. When at 25 cmaway an object appears clearest and this is known as least distance of vision or near point.
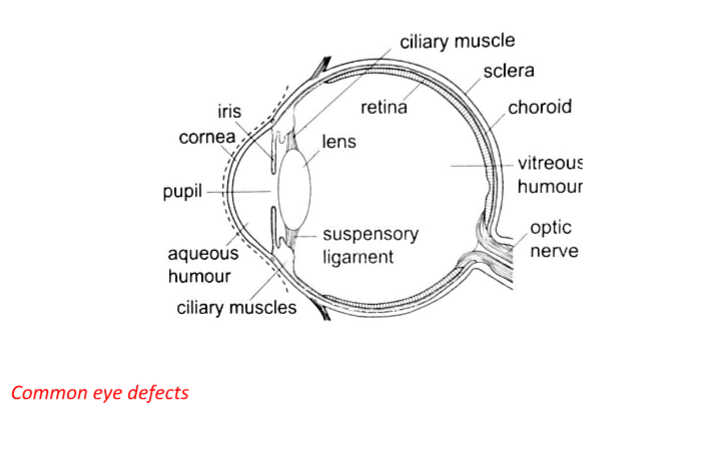
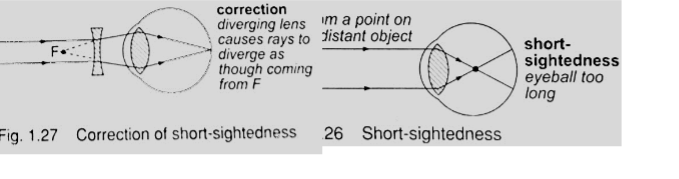
1. Short sightedness or hypermetropia– the eyeball is too large for the ‘relaxed focal length’ of the eye. The defect is corrected by placing a concave lens in front of the eye.
4. Astigmatism – this is a defect where the eye has two different focal lengths as a result of the cornea not being spherical. Corrected by the use of cylindrical lens.
5. Colour blindness– caused by deficiency of colour detecting cells in the retina.
Power of lens
The power of a simple lens is given by the formula: Power = 1 / f. The unit for power of a lens is diopter (D).
Example
Find the power of a concave lens of a focal length 25 cm.
Solution
Power = 1 / f = 1 / -0.25 = -4 D.
Chapter Two
Uniform Circular Motion
Introduction
Circular motion is the motion of bodies travelling in circular paths. Uniform circular motion occurs when the speed of a body moving in a circular path is constant. This can be defined as motion of an object at a constant speed along a curved path of constant radius. When acceleration (variation of velocities) is directed towards the centre of the path of motion it is known as centripetal acceleration and the force producing this centripetal acceleration which is also directed towards the centre of the path is called centripetal force.
This motion can be described as the motion of a body moving along a circular path by giving the angle covered in a certain time along the path of motion.
The angle covered in a certain time is proportional to the distance covered along the path of motion.
The radian
One radian is the angle subtended at the centre of the circle by an arc of length equal to the radius of the circle. Since one circle = 3600and has 2 π radians therefore 1 radian = 3600 / 2 π r= 57.2960 or 57.30.
Example
A wheel of radius 50 cm is rolled through a quarter turn. Calculate
(i) The angle rotated in radians
(ii) The distance moved by a point on the circumference.
Solution
(i) A quarter turn = 3600 / 4= 900. Since 3600 = 2 π radians.
Alternately since 1 radian = 57.30 hence 900 = 1.57 radii.
(ii) A point on the circumference moves through an arc,
Arc = radius ×θ (θ in radians)
= 50 cm × 1.57
= 78.5 cm.
Angular velocity
If a body moving in a circular path turns through an angle θ radians in time ‘t’, we define angular velocity omega (ω), as the rate of change of the angle θ with time.
ω= θ / t, unit for angular velocity is radians per second (rads-1).
Since the radian measure is a ratio we can write it as second-1 (s-1). We can establish the relationship between angular velocity ‘ω’ and linear velocity ‘v’, from the relation, θ = arc / radius, arc = radius × θ.Dividing the expression by ‘t’, then arc / t = radius, but arc / t = v (angular velocity). So ‘v’ = radius × ω. This expression gives us the relationship between angular and linear velocity.
Angular acceleration
If the angular velocity for a body changes from ‘ω1’ to ‘ω2’, in time ‘t’ then the angular acceleration, α can be expressed as;
α= (ω2 – ω1) / t
Units for angular acceleration are radians per second squared (rad s-2) or second-2 (s-2). When α is constant with time, we say the body is moving with uniform angular acceleration. Note: In uniform circular motion α is equal to zero.
To establish the relationship between angular acceleration and linear acceleration, from the relation, v = radius × ω, then dividing by ‘t’, we get (v / t) = radius × ω / t. But v / t = a (linear acceleration) and ω / t = α (angular acceleration). So a = radius × α.
Centripetal force.
This is a force which acts on a body by directing the body towards its centre. Since the direction is continuously changing, the velocity therefore cannot be constant.
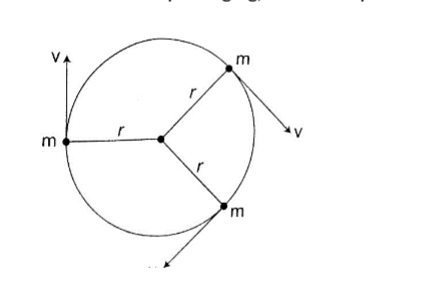
Fc = ma = mv2/R. Since v = radius ω, then
Fc = mv2 – ω2/R = mRω2.
The centripetal acceleration ‘a’ in relation to angular velocity, ω, is given by a = Rω2.
Motion in a vertical circle
Consider a mass ‘m’ tied to a string of length ‘r’ and moving in a vertical circle as shown below.
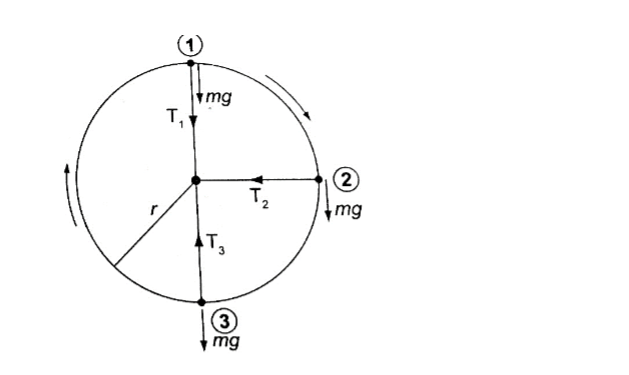
T1 = mv2/r – mg.(The velocity decreases as T1decreases since mg is constant).
T1will be zero when mv2/r = mg and thus v = – this is the value of minimum speed at position 1 which keeps the body in a circle and at this time when T = 0 the string begins to slacken.
At position 2 – the ‘mg’ has no component towards the centre thus playing no part in providing the centripetal force but is provided by the string alone.
T2 = mv2/r
At position 3 – ‘mg’ and T are in opposite directions, therefore; T3 – mg = mv2/r; T3 = mv2/r + mg– indicates that the greatest value of tension is at T3 or at the bottom of the circular path.
Examples
1. A ball of mass 2.5 × 10-2 kg is tied to a string and whirled in a horizontal circular path at a speed of 5.0 ms-2. If the string is 2.0 m long, what centripetal force does the string exert on the ball?
Solution
Fc = mv2/r = (2.5 × 10-2) × 52 / 2.0 = 0.31 N.
2. A car of mass 6.0 × 103 kg is driven around a horizontal curve of radius 250 m.
if the force of friction between the tyres and the road is 21,000 N. What is the maximum speed that the car can be driven at on a bend without going off the road?
Solution
Fc = force of friction = 21,000, also Fc = mv2/r, hence
21,000 = (6.0 × 103) × v2 / 250, v2 = (21,000 × 250) /6.0 × 103
3. A stone attached to one end of a string is whirled in space in in a vertical plane. If the length of the string is 80 cm, determine the minimum speed at which the stone will describe a vertical circle. (Take g = 10 m/s2).
Solution
Minimum speed v √rg=0.08× 10 = 2.283 m/s.
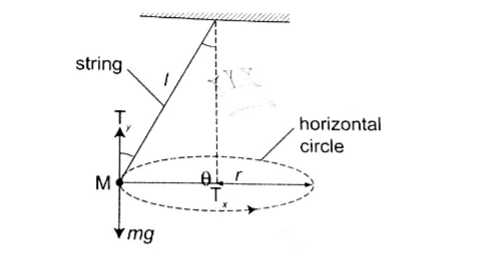
The conical pendulum
It consists of a small massive object tied to the end of a thin string tied to affixed rigid support. The object is then pulled at an angle then made to whirl in a horizontal circle.
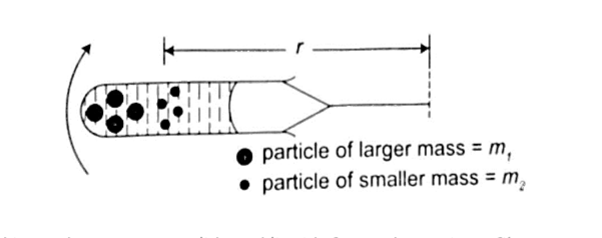
The centrifuge
It consists of a small metal container tubes which can be electrically or manually rotated in a circle. If we consider two particles of different masses m1and m2 each of them requires a centripetal force to keep it in circular motion, the more massive particle require a greater force and so a greater radius and therefore it moves to the bottom of the tube.
Banked tracks
As a vehicle moves round a bend, the centripetal force is provided by the sideways friction between the tyres and the surface, that is;
Centripetal force = mv2/r = frictional force
To enable a vehicle to turn along a bend at high speed the road is raised on the outer edge to attain a saucer-like shape and this is known as banking, where part of the centripetal force necessary to keep the vehicle on track is provided by the weight of the vehicle.
This allows cars to negotiate bends at critical speeds.
Application of uniform circular motion
1. Centrifuges - they are used to separate liquids of different densities i.e. cream and milk
2. Drying clothes in spin dryer- clothes are placed in a perforated drum rotated at high speed, water is expelled through the holes and this makes the clothes dry.
3. Road banking– especially for racing cars which enables them to move at critical speed along bends without going off the tracks.
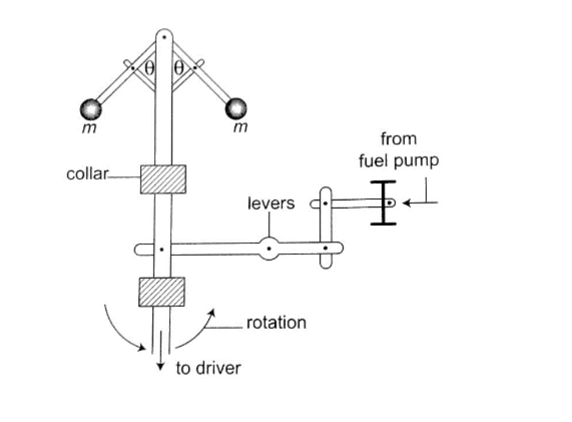
4. Speed governor– the principle of conical pendulum is used here to regulate the speed by controlling the fuel intake in the combustion chamber.
As the collar moves up and down through a system of levers it thereby connects to a device which controls the fuel intake.
Floating and Sinking
Any object in a liquid whether floating or submerged experiences an upward force from the liquid; the force is known as upthrust force.
Upthrust force is also known as buoyant force and is denoted by letter ‘u’.
Archimedes’ principle
Floating and Sinking
Archimedes, a Greek scientist carried out first experiments to measure upthrust on an object in liquid in the third century. Archimedes principle states that ‘When a body is wholly or partially immersed in a fluid (liquid/ gas), it experiences an upthrust equal to the weight of the displaced fluid”.
Floating and Sinking
Experiment: To demonstrate Archimedes principle
Procedure
1. Pour water into an overflow can (eureka can) until it starts to flow out then wait until it stops dripping
2. Tie a suitable solid body securely and suspend it on a spring balance. Determine weight in air.
3. Lower the body slowly into the overflow can while still attached to the spring balance then read off its weight when fully submerged.
4. Weigh the displaced water collected in a beaker.
Record your readings as follows;
Weight of body in air = W1
Weight of body in water = W2
Weight of empty beaker = W3
Weight of beaker and displaced liquid = W4
Upthrust of the body = W1 – W2
Weight of displaced water = W4 – W3
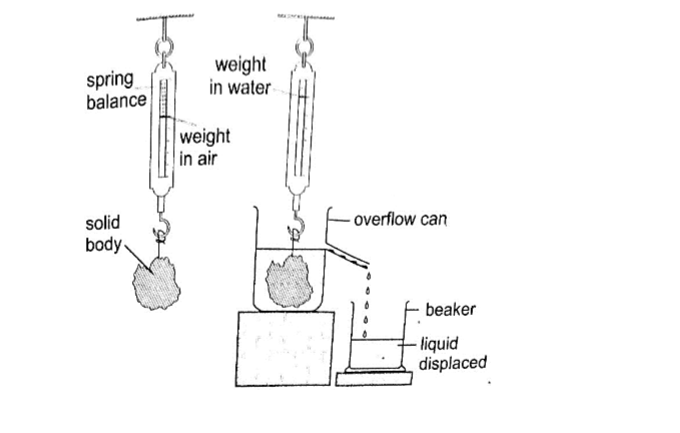
The upthrust on the solid body will be found to be equal to the weight of displaced water therefore demonstrating the Archimedes principle.
Example
A block of metal of volume 60 cm3 weighs 4.80 N in air. Determine its weight when fully submerged in a liquid of density 1,200 kgm-3.
Solution
Volume of liquid displaced = 60 cm3 = 6.0 × 10-5 m3. Weight of the displaced liquid = volume × density × gravity = v × ρ × g
= 6.0 × 10-5 × 1200 × 10
= 0.72 N
Upthrust = weight of the liquid displaced.
Weight of the block in the liquid = (4.80 – 0.72) = 4.08 N.
Floating objects
Objects that float in a liquid are less dense than the liquid in which they float.
We have to determine the relationship between the weight of the displaced liquid and the weight of the body.
Experiment: to demonstrate the law of floatation
Procedure
1. Weigh the block in air and record its weight as W1.
2. Put water into the overflow can (eureka can) up to the level of the spout.
3. Collect displaced water in a beaker. Record the weight of the beaker first in air and record as W2. Weigh both the beaker and the displaced water and record as W3.
4. Record the same procedure with kerosene and record your results as shown below.
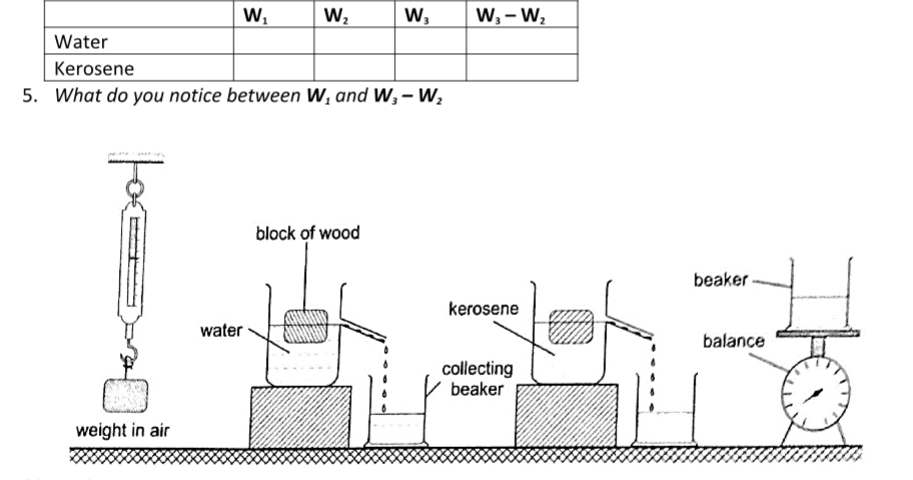
The weight of the displaced liquid is equal to the weight of the block in air.
This is consistent with the law of floatation which states that “A body displaces its own weight of the liquid in which it floats”.
Mathematically, the following relation can be deduced Weight = volume × density × gravity = v × ρ × g, therefore W = v d × ρ × gwhere vdis the volume of displaced liquid.
NOTE – Floatation is a special case of Archimedes principle.
This is because a floating body sinks until the upthrust equals the weight of the body.
Example
A wooden block of dimensions 3 cm × 3 cm × 4 cm floats vertically in methylated spirit with 4 cm of its length in the spirit.
Calculate the weight of the block. (Density of methylated spirit = 8.0 × 102 kgm-3).
Solution
Volume of the spirit displaced = (3 × 3 × 4) = 36 cm3 = 3.6 × 10-5 m3
Weight of the block =v d × ρ × g = (3.6 × 10-5) × 8.0 × 102 × 10 = 2.88 × 10-1 N.
Relative density
We have established the relative density as the ratio of the density of a substance to the density of water. Since by the law of floatation an object displaces a fluid equal to its own weight hence the following mathematical expressions can be established.
Relative density = density of substance / density of water.
= weight of substance / weight of equal volume of water
= mass of substance / mass of equal volume of water
Applying Archimedes principle, the relative density‘d’;
d = weight of substance in air / upthrust in water or d = W / u
Since upthrust is given by (W2 - W1)where W2 – weight in air, W2– weight when submerged. Hence d = W / u = W / W2 – W1, the actual density, ρ of an object can be obtained as follows ρ of an object = d × 1,000 kgm-3.
Relative density of a floating body
Experiment: To determine the relative density of a cork
Procedure
1. Select a sinker which is heavy enough to make the cork to sink.
2. Attach the cork and the sinker as follows.
3. Record the results obtained as follows
Weight of the sinker in water = W1
Weight of the sinker in water and cork in air = W2
Weight of the sinker and cork in water = W3
Weight of the cork in air = W2 – W1
Upthrust on the cork = W2 – W3
The relative density of the cork in air is determined as follows;
d = weight of the cork in air / upthrust on the cork.
Applications of Archimedes principle and relative density
1. Ships – steel which is used to make ships is 6-7 times dense than water but a ship is able to float on water because it is designed to displace more water than its volume.
Load lines called plimsoll marks are marked on the side to indicate the maximum load at different seasons to avoid overloading.
2. Submarines – they are made of steel and consists of ballast tanks which contain water when they have to sink and filled with air when they have to float.
This makes the submarines to balance their weight and be able to rise upwards.
3. Balloons – when they are filled with helium gas balloons become lighter and the upthrust on the balloon becomes greater than their weight therefore becoming able to rise upwards.
4. Hydrometers – they are used to measure the relative densities of liquids quickly and conveniently. Various types of hydrometers are made to measure different ranges of different densities i.e. lactometer – for measuring milk water (range 1.015 – 1.045), battery acid tester – used to test the charge in a lead-acid battery.
Examples
1. A solid of mass 1.0 kg is suspended using a thread and then submerged in water.
If the tension on the thread is 5.0 N, determine the relative density of the solid.
Solution
Mass of solid = 1.0 kg
Weight of solid W = mg = 10 N
Tension on the string (T) = 5 N
Upthrust on solid (u) = W – T = 10 – 5 = 5
Relative density (d) = W / u = 10 / 5 = 2.
2. A balloon made up of a fabric weighing 80 N has a volume of 1.0 × 107 cm. the balloon is filled with hydrogen of density 0.9 kgm-3.
Calculate the greatest weight in addition to that of the hydrogen and the fabric, which the balloon can carry in air of average density 1.25 kgm-3.
Solution
Upthrust = weight of the air displaced
= volume of air × density × gravity
= (1.0 × 107 × 106) × (1.25 × 10)
= 10 × 1.25 × 10 = 125 N
Weight of hydrogen = 10 × 0.09 × 10 = 9 N
Total weight of hydrogen and fabric = 80 + 9 = 89 N
Total additional weight to be lifted = 125 – 89 = 36 N.
3. A material of density 8.5 gcm-3 is attached to a piece of wood of mass 100g and density 0.2 gcm-3.
Calculate the volume of material X which must be attached to the piece of wood so that the two just submerge beneath a liquid of density 1.2 gcm-3.
Solution
Let the volume of the material be V cm3
The mass of the material be 8.5 V grams
Volume of wood = 100 g / 0.2 g/cm = 500 cm3.
In order to have an average density of 1.2 gcm-3 = total mass / total volume
Therefore (100 + 8.5V) / (500 + V) = 1.2 gcm-3
Hence V = 68.5 cm3.
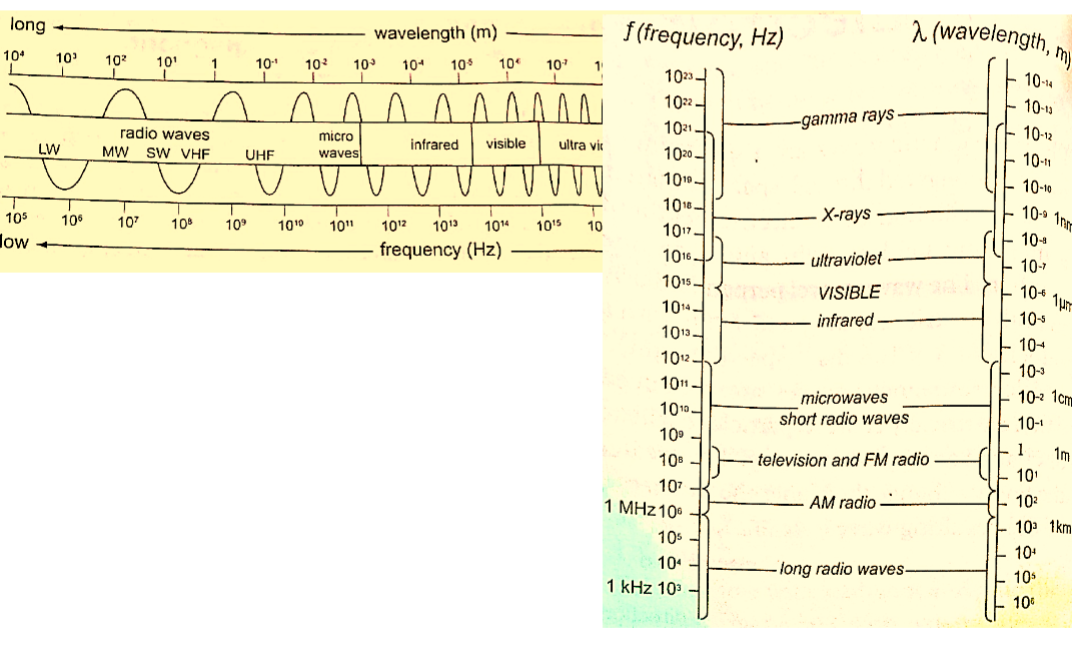
Electromagnetic spectrum is a continuum of all electromagnetic waves arranged according to frequency and wavelength.
It includes visible light, ultra-violet rays, microwaves, X-rays, radio waves and gamma rays.
Electromagnetic waves are produced when electrically charged particles oscillate or change energy in some way. The waves travel perpendicularly to both electric and magnetic fields.
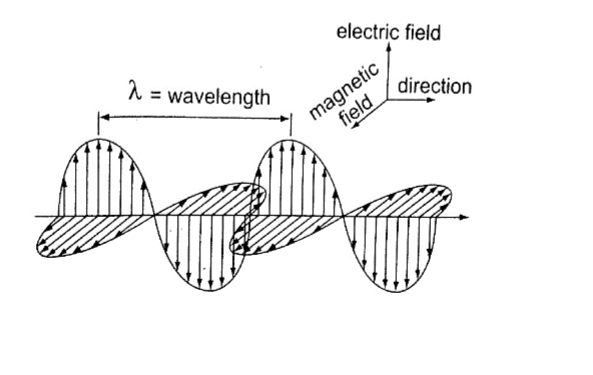
X-rays and gamma rays are usually described in terms of wavelength and radio waves in terms of frequency.
The electromagnetic spectrum
It is divided into seven major regions or bands. A band consists of a range of frequencies in the spectrum in terms of frequencies i.e. radio, microwaves, infra-red.
Properties of electromagnetic waves
Common properties
i. They do not require a material medium and can travel through a vacuum.
ii. They undergo reflection, refraction and diffraction.
iii. All electromagnetic waves travel at the speed of light i.e. 3 × 108 ms-1.
iv. They carry no electric charge
v. They transfer energy from a source to a receiver in the form of oscillating electric and magnetic fields.
vi. They obey the wave equation (v = λ f).
1. A VHF radio transmitter broadcasts radio waves at a frequency of 30 M Hz. What is their wavelength?
Solution
v = f λ => then λ = v / f = 3.0 × 108 / 300 × 106 = 1.00 m.
2. Calculate the frequency of a radio wave of wavelength 150 m.
Solution
v = f λ =>f = v / λ = 2.0 × 106 = 2 M Hz.
Unique properties
1. Radio waves – they are further divided into long waves (LW), medium waves (MW) and short waves (SW).
They are produced by electrical circuits called oscillators and they can be controlled accurately.
They are easily diffracted by small objects like houses but not by large objects like hills.
2. Microwaves – they are produced by oscillation of charges in special aerials mounted on dishes. They are detected by special receivers which convert wave energy to sound i.e. ‘RADAR’ – Radio Detection and Raging.
3. Infra-red radiation – infra-red radiations close to microwaves are thermal (produce heat) i.e. sun, fire but those closer to the visible light have no thermal properties i.e. TV remote control system. Detectors of infra-red radiation are the human skin, photographic film etc.
4. Optical spectrum (visible light) – they form a tiny part of the electromagnetic spectrum. Sources include the sun, electricity, candles etc. these have wavelengths visible to the human eye and includes the optical spectrum (ROYGBIV). It is detected through the eyes, photographic films and photocells.
5. Ultra-violet rays (UV) – has shorter wavelength than visible light. It is emitted by very hotobjects i.e. the sun, welding machines etc. Exposure to UV rays may cause skin cancer and cataracts. They can be detected through photographic film.
6. X-rays – they have very short wavelength but are high energy waves. They are produced in X-ray tubes when high speed electrons are stopped by a metallic object. They are detected by the use of a photographic film or a fluorescent screen.
7. Gamma rays – produced by some radioactive materials when large changes of energy occur inside their nuclei.
They can be detected by the use of photographic films, Geiger Muller tube or a cloud chamber.
Applications of electromagnetic radiation
1. Radio waves – they are used in radio, TV and cellular mobile communications. -Used in military communications (satellite imagery) to form an image of the ground even when there are clouds.
2. Microwaves - used in radar communications by giving direction and distance.
-Used in speed guns by the police to detect over speeding.
-Used in microwave ovens to warm food. The food becomes warm by absorbing energy.
-Used reliably for communication (telephone and computer data).
3. Infra-red radiation - used to produce images of hot objects through the colours
-Produced by the amount of heat dissipated by an object.
-Images produced by satellites give important information on vegetation cover in all areas of the globe. They can also detect fires.
-They are used in hospitals to detect illnesses (diagnosis)
-Used in warfare missiles and burglar alarm systems
-Used in green houses to grow crops
4. Visible light - used by plants in remote sensing and humans in the identification of things
-Used by plants in the process of photosynthesis
5. Ultra-violet (UV) radiation – used to make reflective materials which absorb light and re-emit it as visible light.
-Used in banks to detect fake currency
6. X-rays - used in hospitals to detect fractures, broken bones and in treatment of cancer (radiotherapy).
-Used to detect foreign materials in the body i.e. metals
-Used to detect invisible cracks in metal castings and welding joints
7. Gamma rays - used to sterilize medical instruments -Used to kill weevils in grain
-Used to take photographs same way like X-rays.
Chapter Five
Electromagnetic Induction
Electromagnetism is the effect resulting from the interaction between an electric current and a magnetic field. This effect brings about induced electromagnetic force (e.m.f) and the resulting current is called induced current.
Experiments on electromagnetic induction
Consider the following diagram
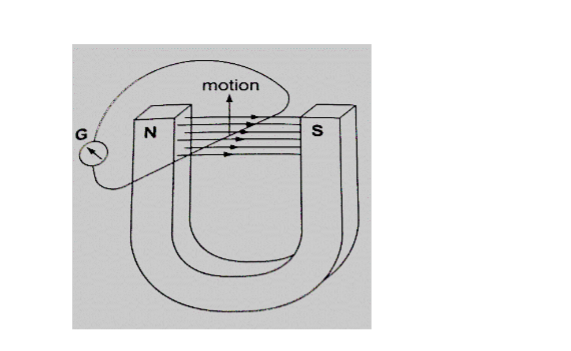
When moved horizontally or held in a fixed position there is no deflection in the galvanometer.
This shows that e.m.f is induced due to the relative motion of the wire or the magnet.
Factors affecting the magnitude of the induced e.m.f
1. The rate of relative motion between the conductor and the field – if the velocity of the conductor is increased the deflection in the conductor increases.
2. The strength of the magnetic field – a stronger magnetic field creates a bigger deflection
3. The length of the conductor – if the length is increased in the magnetic field the deflection increases.
Faraday’s law of magnetic induction
After considering the factors affecting the magnitude of the induced e.m.f, Michael Faraday came up with a law which states that “The induced e.m.f in a conductor in a magnetic field is proportional to the rate of change of the magnetic flux linking the conductor”.
Lenz’s law of electromagnetic induction
This law is used to determine the direction of the induced current in a conductor. It states that “An induced current flows in such a direction that its magnetic effect opposes the change through which the current has been produced”.
It is applied similarly when a wire is been moved in magnetic field.
Fleming’s right hand rule.
The law states that “The first finger, the second finger and the thumb of the right hand when placed mutually perpendicular to each other, the first finger points in the direction of the field and the thumb in the direction of motion then second finger points in the direction of the induced current”. This law is also called the generator rule.

1. A.c generator/alternator – a generator is a device which produces electricity on the basis of electromagnetic induction by continuous motion of either a solenoid or a magnet.
It consists of an armature made of several turns of insulated wire wound on soft iron core and revolving freely on an axis between the poles of a powerful magnet.
Two slip rings are connected to the ends of the armature with two carbon brushes rotating on the slip ring.
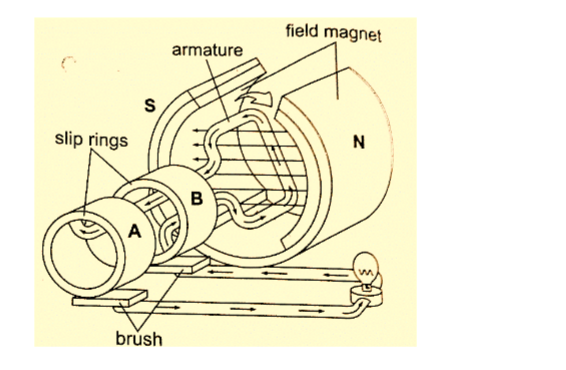
This brings about alternating current and the corresponding voltage (e.m.f) is the alternating voltage. They are used in car alternators and H.E.P.
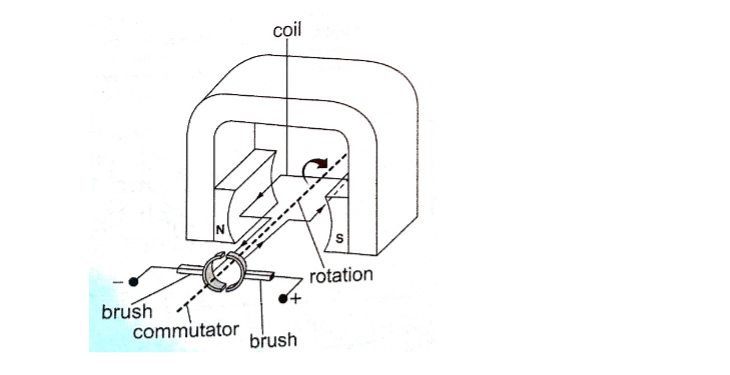
2. D.c generator/alternator – in this case the commutators replaces the slip rings to enable the output to move in one direction.
After a rotation of 1800, instead of current reversing, the connections to the external circuit are reversed so that current direction flows in one direction.
Eddy currents
They are composed of loops of current which have a magnetic effect opposing the force producing them.
When a copper plate with slits is used the loops are cut off and hence the effective currents are drastically reduced and so is the opposing force.

The lamination slices, which are quite thin are glued together by a non-conducting glue and this reduces eddy currents to an almost negligible value.
Eddy currents are useful in moving coil meters to damp the oscillations of the armature when the current is switched off.
Mutual induction
Mutual induction is produced when two coils are placed close to each other and a changing current is passed through one of them which in turn produces an induced e.m.f in the second coil. Therefore mutual induction occurs when a changing magnetic flux in one coil links to another coil.
Applications of mutual induction
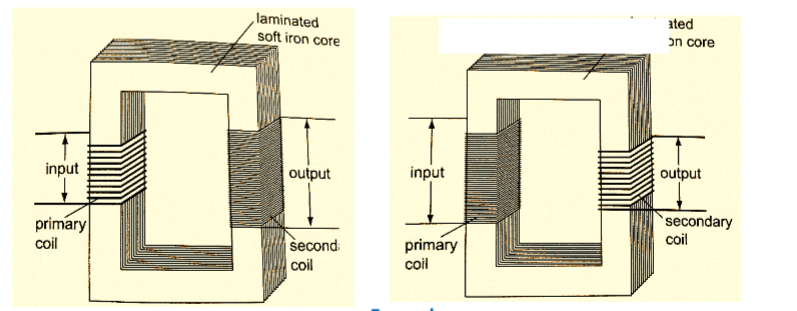
1. The transformer - it converts an alternating voltage across one coil to a larger or smaller alternating voltage across the other. Since H.E.P is lost through transmission lines therefore it is stepped down before it being transmitted and stepped up again at the point of supply lines.
In a step up transformer the number of turns in the secondary coil (Ns) is higher than the number of turns in the primary coil (Np).
In a step down transformer the primary coil has more turns than the secondary coil.
The relationship between the primary voltage and the secondary voltage is given by;
Np / Ns = Vp / Vs.The efficiency of a transformer is the ratio of power in secondary coil (Ps) to power in primary coil (Pp), therefore efficiency = Ps / Pp × 100%.
1. A current of 0.6 A is passed through a step up transformer with a primary coil of 200 turns and a current of 0.1 A is obtained in the secondary coil. Determine the number of turns in the secondary coil and the voltage across if the primary coil is connected to a 240 V mains.
Solution
Np / Ns = Vp / Vs = Ip / Is = Ns = (0.6 × 200) / 0.1 = 1200 turns
Vp = 240 V hence Vs = (240 × 1200) / 200 = 1440 V
2. A step-up transformer has 10,000 turns in the secondary coil and 100 turns in the primary coil.
An alternating current of 0.5 A flows in the primary circuit when connected to a 12.0 V a.c. supply.
a) Calculate the voltage across the secondary coil
b) If the transformer has an efficiency of 90%, what is the current in the secondary coil?
Solution
a) Vs = (Ns / Np) × Vp = (10,000 × 12) / 100 = 1200 V
b) Power in primary = Pp = Ip × Vp= 5.0 × 12 = 60 W
Efficiency = Ps / Pp × 100% but Ps = Is Vs
Is = (60 × 90) / (1200 × 100) = 0.045 A
Energy losses in a transformer.
Loss of energy in a transformer is caused by;
i) Flux leakage– this may be due to poor transformer design
ii) Resistance in the windings–it is reduced by using copper wires which have very low resistance
iii) Hysteresis losses– caused by the reluctance of the domains to rotate as the magnetic field changes polarity. Reduced by using materials that magnetize and demagnetize easily like soft iron in the core of the transformer.
iv) Eddy currents– reduced by using a core made of thin, well insulated and laminated sections.
Uses of transformers
1. Power stations – used to step up or down to curb power losses during transmission
2. Supplying low voltages for school laboratories
3. Low voltage supply in electronic goods like radios, TVs etc.
4. High voltage supply in cathode ray oscilloscope (CRO) for school laboratories.
3. Induction coil –was developed in 1851 by Heinrich Ruhmkortt. It has both secondary and primary coils with an adjustable spark gap.
4. Car ignition system – it is applied in petrol driven engines where a spark plug is used to ignite petrol vapour and air mixture to run the engine.
Chapter Six
Mains Electricity
Sources of mains electricity
Mains electricity comes from a power station and its current is the alternating current which can either be stepped up or down by a transformer.
A.c is produced when a conductor is rotated in a magnetic field or when a magnetic field is rotated near a conductor.
This method is known as electromagnetic induction. The source of energy for rotating the turbine is the actual source of electrical energy.
Most of the electricity in East Africa is generated from water.
Power transmission
This is the bulk transfer of electric power from one place to another. A power transmission system in a country is referred to as the national grid.
This transmission grid is a network of power generating stations, transmission circuits and sub-stations. It is usually transmitted in three phase alternating current.
Grid input
At the generating plant the power is produced at a relatively low voltage of up to 25 kV then stepped up by the power station transformer up to 400 kV for transmission. It is transmitted by overhead cables at high voltage to minimize energy losses.
The cables are made of aluminium because it is less dense than copper.
Metallic poles (pylons) carry four cables, one for each phase and the fourth is the neutral cable which is thinner and completes the circuit to the generator.
Grid exit
At sub-stations transformers are used to step down voltage to a lower voltage for distribution to industrial and domestic users.
The combination of sub-transmission (33 kV to 132 kV) and distribution (11 kV to 33 kV) which is then finally transformed to a voltage of 240 V for domestic use.
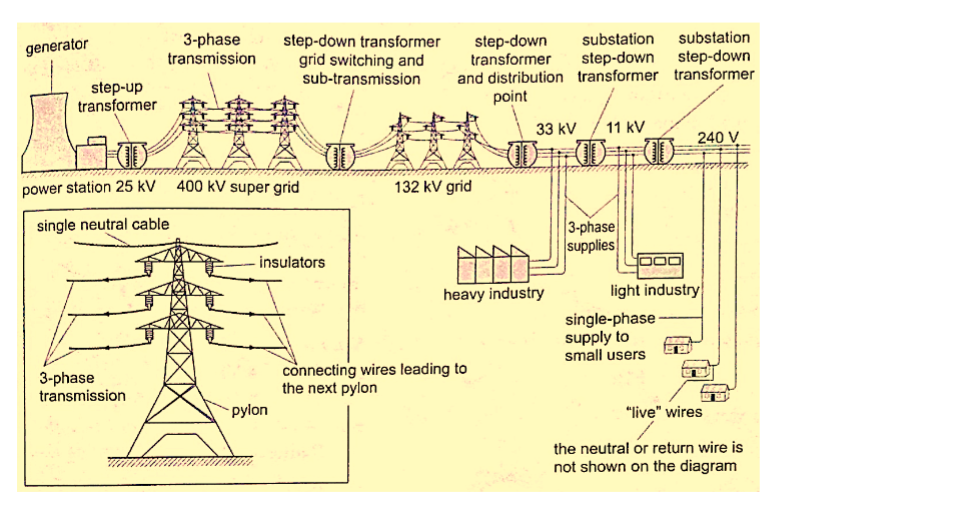
This is the penultimate process of delivery of electric power. It is considered to include medium voltage (less than 50 kV) power lines, low voltage (less than 1,000 V) distribution, wiring and sometimes electricity meters.
Dangers of high voltage transmission
1. They can lead to death through electrocution
2. They can cause fires during upsurge
3. Electromagnetic radiations from power lines elevate the risk of certain types of cancer
Electrical power and energy
Work done = volts × coulombs = VQ, but Q = current × time = I t.
So work done = V I t
Other expressions for work may be obtained by substituting V and I from Ohms law as below V = I R and I = V / R, work done = I R × I t = I2 R t, or work done = V × V t / R = V2 t / R. The three expressions can be used to calculate work done. Electrical power may be computed from the definition of power. Power = work / time = I2 R t /t = I2 R or V2 t / R t = V2 / R Using work done = V I t, then Power = V I.
These expressions are useful in solving problems in electricity. Work done or electrical energy is measured in joules (J) and power is measured in watts (W). 1 W = 1 J/s.
Example
An electric heater running on 240 V mains has a current of 2.5 A.
a) What is its power rating?
b) What is the resistance of its element?
Solution
a) Power = V I = 240 × 2.5 = 600 W. Rating is 600 W, 240 V.
b) Power = V / R = 600 W. R = V / I. R = 240 / 2.5 = 96 Ω.
Costing electricity
The power company uses a unit called kilowatt hour (kWh) which is the energy transformed by a kW appliance in one hour.
1 kW = 1,000 W × 60 × 60 seconds = 3,600,000 J. The meter used for measuring electrical energy uses the kWh as the unit and is known as joule meter.
Examples
1. An electric kettle is rated at 2,500 W and uses a voltage of 240 V.
a) If electricity costs Ksh 1.10 per kWh, what is the cost of running it for 6 hrs?
b) What would be its rate of dissipating energy if the mains voltage was dropped to 120 V?
Solution
a) Energy transformed in 6 hrs = 2.5 × 6 = 15 kWh. Cost = 15 × 1.10 × 6 = Ksh 99.00 b) Power = V2 / R = 2500. R = (240 × 240) /2500 = 23.04 Ω.
Current = V / R = (240 × 2500) / (240 × 240) = 10.42 A
Power = V I = (2500 × 120) / 240 = 1,250 W.
2. An electric heater is made of a wire of resistance 100 Ω connected to a 240 V mains supply.
Determine the;
a) Power rating of the heater
b) Current flowing in the circuit
c) Time taken for the heater to raise the temperature of 200 g of water from 230C to 950C. (specific heat capacity of water = 4,200 J Kg-1 K-1)
d) Cost of using the heater for two hours a day for 30 days if the power company charges Ksh 5.00 per kWh.
Solution
a) Power = V2 / R = (240 × 240) / 100 = 576 W
b) P = V I =>> I = P / V = 576 / 240 = 2.4 A
c) P × t = heat supplied = (m c θ) = 576 × t = 0.2 × 4200 × 72.
Hence t = (0.2 × 4200 × 72) / 576 = 105 seconds.
d) Cost = kWh × cost per unit = (0.576 × 2 × 30) × 5.0 = Ksh 172.80
3. A house has five rooms each with a 60 W, 240 V bulb. If the bulbs are switched on fro 7.00 pm to 10.30 pm, calculate the;
a) Power consumed per day in kWh
b) Cost per week for lighting those rooms if it costs 90 cents per unit.
Solution
a) Power consumed by 5 bulbs = 60 × 5 = 300 W = 0.3 kWh. Time = 10.30 – 7.00 = 3 ½ hrs.Therefore for the time duration = 0.3 × 3 ½ = 1.05 kWh.
b) Power consumed in 7 days = 1.05 × 7 = 7.35 kWh. Cost = 7.35 × 0.9 = Ksh 6.62
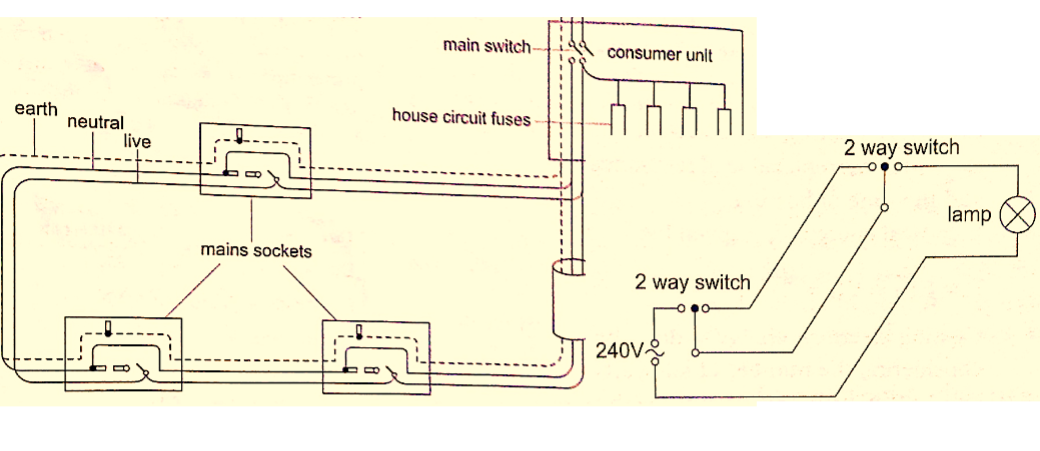
Domestic wiring system
Power is supplied by two cables where one line is live wire (L) and the other is neutral (N). Domestic supply in Kenya is usually of voltage 240 V. The current alternates 50 times per second hence the frequency is 50 Hz.
The neutral is earthed to maintain a zero potential. The main fuse is fitted on the live wire to cut off supply in case of a default. A fuse is a short piece of wire which melts if current of more value flows through it.
Supply to the house is fed to the joule meter which measures the energy consumed.
From the meter both L and N cables go to the consumer box (fuse box) through the main switch which is fitted on the live cable.
Consumer units within the house are fitted with circuit breakers which go off whenever there is a default in the system.
Lights in the house are controlled by a single or double switch (two way).
In most wiring systems the main sockets are connected to a ring main which is a cable which starts and end at the consumer unit. Plugs used are the three- pin type.
Cathode Rays
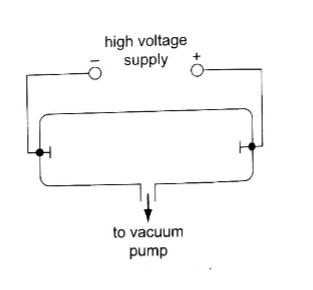
These are streams of electrons emitted at the cathode of an evacuated tube containing an anode and a cathode.
Production of cathode rays
They are produced by a set up called a discharge tube where a high voltage source usually referred to as extra high tension (EHT) supply connected across a tube containing air at low pressure thereby producing a luminous electron discharge between the two brass rods placed at opposite ends of the tube. These electron discharges are called cathode rays which were discovered by J.J Thomson in the 18th century.
1. They travel in straight lines
2. They are particulate in nature i.e. negatively charged electrons
3. They are affected by both magnetic and electric fields since they are deflected towards the positive plates
4. They produce fluorescence in some materials
5. Depending on the energy of the cathode rays they can penetrate thin sheets of paper, metal foils
6. When cathode rays are stopped they produce X-rays.
7. They affect photographic plates.
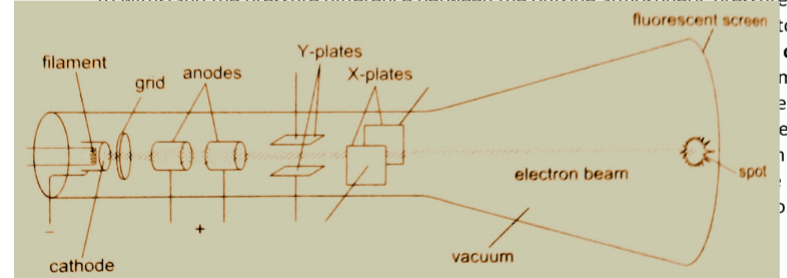
Cathode ray oscilloscope (CRO)
It is a complex equipment used in displaying waveforms from various sources and measuring p.d. It comprises of the following main components; - The cathode ray tubes (CRT) – consists of a tube, electron gun, deflection plates and the time base (TB).
The tube is made of strong glass to withstand the pressure difference between the outside atmospheric pressure and the vacuum inside.
It has a square grid placed in front of it to allow measurements to be made.
The electron gun produces the electrons with main parts consisting of a filament, a cathode, a grid and the anode.
Electrons are produced by the cathode when heated by the filament.
The grid is a control electrode which determines the number of electrons reaching the screen therefore determining the brightness of the screen. The Y-deflection plates deflects the beam up or down.
Clearly observable when low frequency inputs are applied i.e. 10 Hz from a signal operator.
The X-deflection plates are used to move the beam left or right of the screen at a steady speed using the time base circuit which automatically changes voltage to an a.c.
voltage. When time base control is turned the speed can be adjusted to produce a waveform.
1. If the time base control of the CRO is set at 10 milliseconds per cm, what is the frequency of the wave traced given wavelength as 1.8 cm?
Solution
Wavelength = 1.8 cm. time for complete wave = period = 1.8 × 10 milliseconds / cm
= 18 milliseconds
= 1.8 × 10-2 seconds.
Frequency ‘f’, is given by f = 1 / T = 1 / 1.8 × 10-2 = 100 / 1.8 = 56 Hz.
NOTE:
The television set (TV) is a type of a CRT with both Y and X-deflection plates which control the formation of a picture (motion) on the screen.
The colour television screen is coated with different phosphor dots (chemicals) which produce a different colour when struck by an electron beam.
Chapter Eight
X-rays
X-rays were discovered by a German scientist named Roentgen in 1985. They can pass through most substances including soft tissues of the body but not through bones and most metals. They were named X-rays meaning 'unknown rays'.
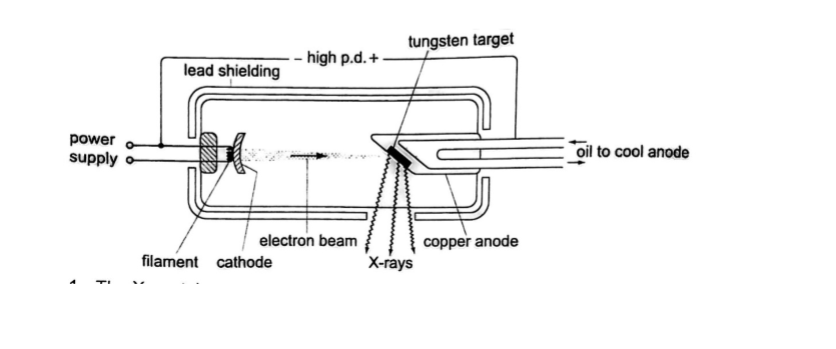
X-ray production
They are produced by modified discharge tubes called X-ray tubes. The cathode is in the form of a filament which emits electrons on heating.
The anode is made of solid copper molybdenum and is called the target. A high potential difference between the anode and the cathode is maintained (10,000 v to 1,000,000 or more) by an external source. The filament is made up of tungsten and coiled to provide high resistance to the current.
The electrons produced are changed into x-rays on hitting the anode and getting stopped.
Only 0.2% of the energy is converted into x-rays.
Cooling oil is led in and out of the hollow of the anode to maintain low temperature. The lead shield absorbs stray x-rays.
When the cathode is heated electrons are emitted by thermionic emission. They acquire electrical energy which can be expressed as E = e V. Once in motion the electrical energy is converted to kinetic energy, that is eV = ½ me v2.
The energy of an electromagnetic wave can be calculated using the following equation Energy = h f,where h- Planck’s constant, f – frequency of the wave.
The highest frequency of the X-rays released after an electron hits the target is when the greatest kinetic energy is lost, that is h f max = eV.
Lower frequencies are released when the electrons make multiple collisions losing energy in stages, the minimum wavelength, λ min, of the emitted X-rays is given by;
(hc) / λ min = eV.
These expressions can be used to calculate the energy, frequencies and wavelengths of X-rays.

i) They travel in straight lines
ii) They undergo reflection and diffraction
iii) They are not affected by electric or magnetic fields since they are not charged particles.
iv) They ionize gases causing them to conduct electricity
v) They affect photographic films
vi) They are highly penetrating, able to pass easily through thin sheets of paper, metal foils and body tissues
vii) They cause fluorescence in certain substances for example barium platinocynide.
Hard X-rays
These are x-rays on the lower end of their range (10-11 – 10-8 m) and have more penetrating power than normal x-rays.
They are capable of penetrating flesh but are absorbed by bones.
Soft X-rays
They are on the upper end of the range and are less penetrative. They can only penetrate soft flesh and can be used toshow malignant growth in tissues.
Dangers of X-rays and the precautions.
1. They can destroy or damage living cells when over exposed.
2. Excessive exposure of living cells can lead to genetic mutation.
3. As a precautionary measure X-ray tubes are shielded by lead shields.
Uses of X-rays
1. Medicine – X-ray photos called radiographs are used as diagnostic tools for various diseases. They are also used to treat cancer in radiotherapy.
2. Industry – they are used to photograph and reveal hidden flaws i.e. cracks in metal casting and welded joints.
3. Science – since the spacing of atomic arrangement causes diffraction of x-rays then their structure can be studied through a process called X-ray crystallography.
4. Security – used in military and airport installations to detect dangerous metallic objects i.e. guns, explosives, grenades etc.
Chapter Nine
Photoelectric Effect
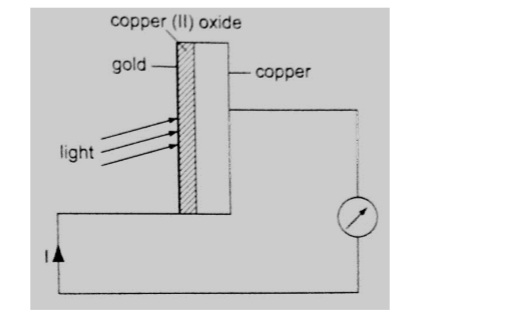
Photoelectric effect was discovered by Heinrich Hertz in 1887. Photoelectric effect is a phenomenon in which electrons are emitted from the surface of a substance when certain electromagnetic radiation falls on it.
Metal surfaces require ultra-violet radiation while caesium oxide needs a visible light i.e. optical spectrum (sunlight).
Work function
A minimum amount of work is needed to remove an electron from its energy level so as to overcome the forces binding it to the surface.
This work is known as the work function with units of electron volts (eV). One electron volt is the work done when one electron is transferred between points with a potential difference of one volt; that is,
1 eV = 1 electron × 1 volt
1 eV = 1.6 × 10-19 × 1 volt
1 eV = 1.6× 10-19 Joules (J)
Threshold frequency
This is the minimum frequency of the radiation that will cause a photoelectric effect on a certain surface. The higher the work function, the higher the threshold frequency.
Factors affecting the photoelectric effect
1. Intensity of the incident radiation – the rate of emission of photoelectrons is directly proportional to the intensity of incident radiation.
2. Work function of the surface – photoelectrons are emitted at different velocities with the maximum being processed by the ones at the surface.
3. Frequency of the incident radiation – the cut-off potential for each surface is directly proportional to the frequency of the incident radiation.
Planck’s constant
When a bunch of oscillating atoms and the energy of each oscillating atom is quantified i.e. it could only take discrete values.
Max Planck’s predicted the energy of an oscillating atom to be E = n h f, where n – integer, f – frequency of the source, h – Planck’s constant which has a value of 6.63 × 10-34 Js.
Quantum theory of light
Planck’s published his quantum hypothesis in 1901 which assumes that the transfer of energy between light radiation and matter occurs in discrete units or packets.
Einstein proposed that light is made up of packets of energy called photons which have no mass but they have momentum and energy given by;
E = h f
The number of photons per unit area of the cross-section of a beam of light is proportional to its intensity. However the energy of a photon is proportional to its frequency and not the intensity of the light.
Einstein’s photoelectric equation
As an electron escapes energy equivalent to the work function ‘Φ’ of the emitter substance is given up. So the photon energy ‘h f’ must be greater than or equal to Φ. If the ‘h f’ is greater than Φ then the electron acquires some kinetic energy after leaving the surface.
The maximum kinetic energy of the ejected photoelectron is given by;
K.E max = ½ m v2max = h f – Φ ……………… (i), where m v2max = maximum velocity and mass.
This is the Einstein’s photoelectric equation.
If the photon energy is just equivalent to work function then, m v2max = 0, at this juncture the electron will not be able to move hence no photoelectric current, giving rise to a condition known as cut-off frequency, h fco = Φ………………. (ii)
Also the p.d required to stop the fastest photoelectron is the cut-off potential, V cowhich is given by E = e V co electron volts, but this energy is the maximum kinetic energy of the photoelectrons and therefore, ½ m v2max = e V co ………….. (iii).
Combining equations (i), (ii) and (iii), we can write Einstein’s photoelectric equation as, e V co = h f – h fco ………………….. (iv)
NOTE: -- Equations (i) and (iv) are quite useful in solving problems involving photoelectric effect.
Examples
1. The cut-off wavelength for a certain material is 3.310 × 10-7 m. What is the cut-off frequency for the material?

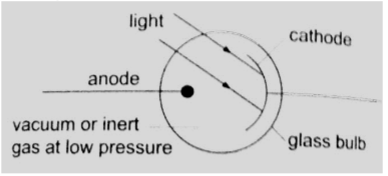
1. Photo-emissive cells – they are made up of two electrodes enclosed in a glass bulb (evacuated or containing inert gas at low temperature).
The cathode is a curved metal plate while the anode is normally a single metal rod)
Other devices such as photo-diodes and photo-transistors block current when the intensity of light increases.
Photo-conductive cells are also known as light dependent resistors (LDR) and are used in alarm circuits i.e. fire alarms, and also in cameras as exposure metres.
Chapter Ten
Radioactivity
Introduction
Radioactivity was discovered by Henri Becquerel in 1869. In 1898, Marie and Pierre Curie succeeded in chemically isolating two radioactive elements, Polonium (z=84) and Radium (z= 88).
Radioactivity or radioactive decay is the spontaneous disintegration of unstable nuclides to form stable ones with the emission of radiation.
Unstable nuclides continue to disintegrate until a stable atom is formed.
Alpha (α) and beta (ϐ) particles are emitted and the gamma rays (ϒ) accompany the ejection of both alpha and beta particles.
The nucleus
The nucleus is made up of protons and neutrons.
They are surrounded by negatively charged ions known as electrons.
The number of protons is equal to the number of electrons. Both protons and neutrons have the same mass.
The weight of an electron is relatively small compared to neutrons and protons.
The number of protons in an atom is referred to as the proton number (atomic number) and denoted by the symbol Z.
the number of neutrons is denoted by the symbol N. Protons and neutrons are called nucleons since they form the nucleus of an atom.
The sum of both the protons and neutrons is called the mass number A or nucleon number.
Therefore;
A = Z + N and N = A – Z.
The masses of atoms are conveniently given in terms of atomic mass units (ᴜ) where (ᴜ) is 1/12th the mass of one atom of carbon-12 and has a value of 1.660 × 10-27 kg.
Hence the mass of one proton is equal to 1.67 × 10-27 and is equal to 1ᴜ.
Radioactive isotopes
Isotopes are elements with different mass numbers but with equal atomic numbers i.e. uranium with mass numbers 235 and 238.
Properties of radioactive emissions
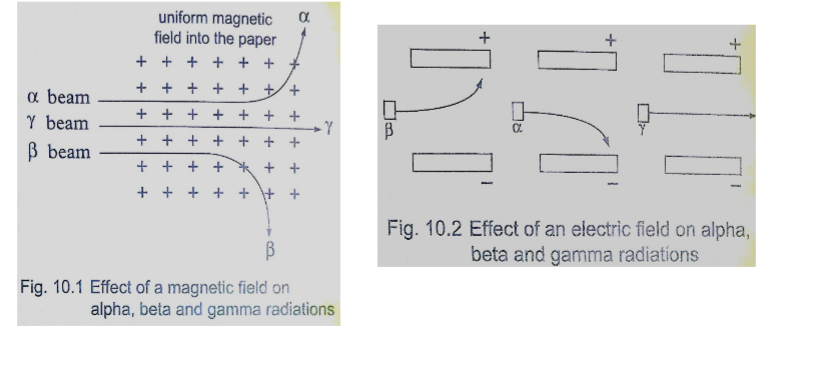
a) Alpha (α) particles
They are represented as , hence with a nucleus number 4 and a charge of +2. Properties
1. Their speeds are 1.67 × 107 m/s, which is 10% the speed of light.
2. They are positively charged with a magnitude of a charge double that of an electron.
3. They cause intense ionization hence loosing energy rapidly hence they have a very short range of about 8 cm in air.
4. They can be stopped by a thin sheet of paper, when stopped they capture two electrons and become helium gas atoms.
5. They can be affected by photographic plates and produce flashes when incident on a fluorescent screen and produce heating effect in matter.
6. They are slightly deflected by a magnetic field indicating that they have comparatively large masses.
b) Beta (ϐ) particles
They are represented by meaning that they have no mass but a charge of -1. Properties
1. Their speeds are as high as 99.9% or more the speed of light
2. They are deflected by electric and magnetic fields but in a direction opposite to that of alpha particles.
3. Due to their high speed they have a higher penetrative rate than alpha particles (about 100 times more)
4. They can be stopped by a thin sheet of aluminium
5. Their ionization power is much less intense about 1/100th that of alpha particles. c) Gamma (ϒ) particles
They have very short wavelengths in the order of 10-10 m and below.
Properties
1. They travel at the speed of light.
2. They have less ionization power than that of both alpha and beta particles
3. They accompany the emission of alpha and beta particles
4. They carry no electric charge hence they are not deflected by both electric and magnetic fields.
5. They have more penetrating power than X-rays.
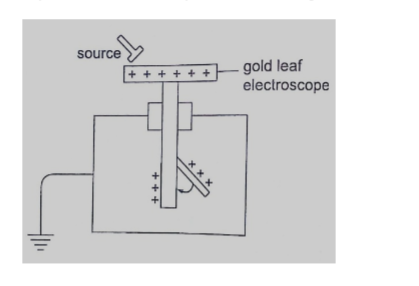
1. Gold leaf electroscope–the rate of collapse of the leaf depends on the nature and intensity of radiation.
The radioactive source ionizes the air around the electroscope. Beta particles discharges a positively charged electroscope with the negative charge neutralizing the charge of the electroscope. Alpha particles would similarly discharge a negatively charged electroscope.
To detect both alpha and beta particles a charged electroscope may not be suitable because their ionization in air may not be sufficiently intense making the leaf not to fall noticeably.
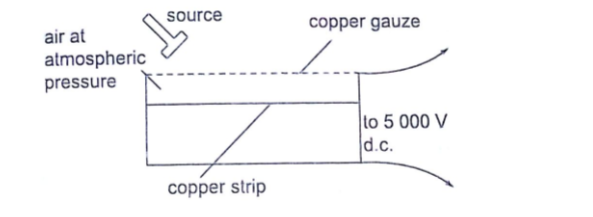
By putting the source away from the gauze or placing a sheet of paper between the two one can determine the range and penetration of the alpha particles.
3. Geiger Muller (GM) tube– it is illustrated as below
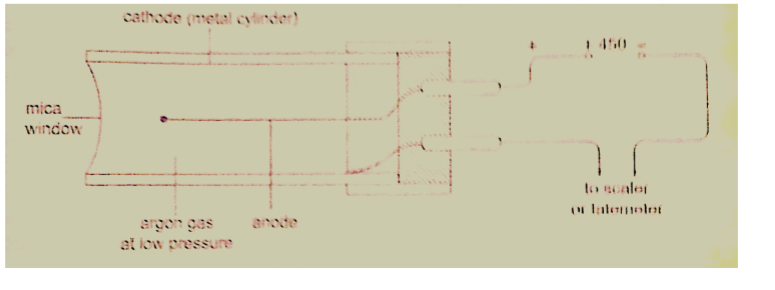
The radiations ionize the gas inside the tube. The electrons move to the anode while the positive ions move to the cathode. As the ions are produced there are collisions which produce small currents which are in turn amplified and passed to the scale.
The scale counts the pulses and shows the total on a display screen.
After each pulse the gas returns to normal ready for the next particle to enter.
A small presence of halogen gas in the tube helps in absorbing the positive ions to reduce further ionization and hence a quick return to normal. This is called quenching the tube.
4. The solid state detector– this detector can be used to detect alpha, beta and gamma radiations where the incoming radiation hits a reverse biased p-n junction diode momentarily conducting the radiation and the pulse of the current is detected using a scaler.
5. The diffusion cloud chamber – this chamber is simplified as shown below
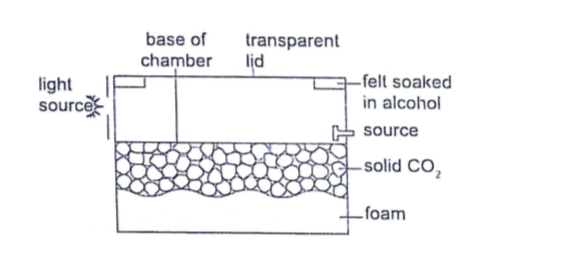
It is cooled below its normal condensing temperature.
As a particle enters the chamber it ionizes the air in its path and alcohol condenses around the path to form millions of tiny alcohol droplets leaving a trail visible because it reflects light from the source.
Alpha particles leave a thick, short straight tracks.
Beta particles leave thin irregular tracks.
Gamma particles do not produce tracks and since they eject electrons from atoms the tracks are similar to those of beta particles.
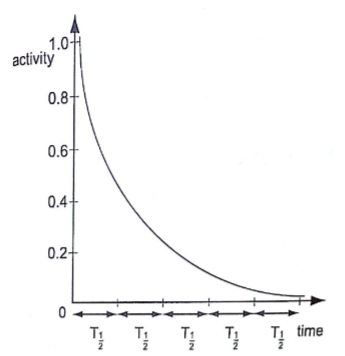
Activity and half-life of elements
The activity of a sample of radioactive element is the rate at which its constituent nuclei decay or disintegrate.
It is measured in disintegrations per second or Curie (Ci) units, where 1 Ci = 3.7 × 1010 disintegrations per second
1 micro Curie (µ C) = 3.7 × 104 disintegrations per second.
The law of radioactive decay states that “the activity of a sample is proportional to the number of undecayed nuclei present in the sample”.
The half-life of a radioactive element is the time required for its one-half of the sample to decay.
It is important to note that although the activity approaches zero, it never goes to zero.
1. The half-life of a sample of a radioactive substance is 98 minutes.
How long does it take for the activity of the sample to reduce to 1/16th of the original value?

Solution
a) 2,400 × ½ × ½ × ½ = 300
Three half-lives have a total of 30 hours, thus half-life = 30 /3 = 10 hours
b) Since half-life = 10 hrs half-lives in 50 hrs = 50/10 = 5 hrs.
So the remaining undecayed atoms are ½ × ½ × ½ × ½ × ½ × 6.0 × 1020
= 0.1875 × 1020, thus
The number of atoms which have decayed = (6.0 – 0.1875) × 1020
= 5.812 × 1020
Nuclear equations
Particles making an atom can be written using upper and lower subscripts where a proton, ‘p’ with charge +1 and mass 1ᴜ, is written as .
A neutron ‘n’ with no charge but with mass 1ᴜ, is written as , while an electron with a charge of -1 and negligible mass is written as . It is important to note that the principles of conservation apply in radioactive decay.
That means that the total number of nucleons (neutrons + protons) must be the same before and after decay. The L.H.S of the equation must be equal to the R.H.S for both total mass and charge.
Effects of radioactive decay on the nucleus


Nuclear fission is a process in which a nucleus splits into two or more lighter nuclei. This process generates large amounts of energy together with neutron emission. Nearly 80% of the energy produced appears as kinetic energy of the fission fragments.
For example Uranium-235 undergoes nuclear fission when bombarded with slow neutrons releasing 2-3 neutrons per Uranium molecule and every neutron released brings about the fission of another Uranium-235nuclei.
Another substance which undergoes the same process is Plutonium-239.
Substances which undergo fission directly with slow neutrons are known as fissile substances or isotopes.
Applications of nuclear fission
1. They are used in the manufacture of atomic bombs where tremendous amount of energy is released within a very short time leading to an explosion.
2. When this release of energy is controlled such that it can be released at a steady rate then it is converted into electrical energy hence the principle in nuclear reactors.
Nuclear fusion
Nuclear fusion is the thermal combining of light elements to form relatively heavier nuclei. The process requires very high temperatures for the reacting nuclei to combine upon collision.
These temperatures are provided by ordinary fission bombs.
These reactions sometimes known as thermonuclear reactions.
A fusion reaction releases energy at the rate of 3-23 MeV per fusion event i.e. two deuterium (heavy hydrogen) nuclei to form helium.
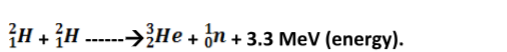
Application of nuclear fusion
1. Used in the production of hydrogen bomb. Possible reactions for an hydrogen bomb include;
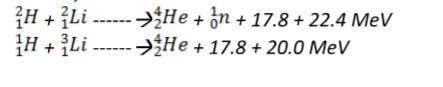
(i) Due to the ionizing radiation emitted by radiation materials, they affect living cells leading to serious illnesses. Symptoms of radiation exposures are immature births, deformations, retardedness, etc.
(ii) Their exposure to the environment through leaks may lead to environmental pollution leading to poor crop growth and destruction of marine life.
Applications of radioactivity
1. Carbon dating – through the identification of carbon-14 and carbon-12 absorbed by dead plants and animals. Scientists can be able to estimate the age of a dead organism. Since carbon is a radioactive element with a half-life of 5,600 years archeologists can be able to estimate the ages of early life through carbon dating.
2. Medicine – radiation is used in the treatment of cancer, by using a radioactive cobalt-60 to kill the malignant tissue. Radiations are used in taking x-ray photographs using cobalt-60. Radiations are used to sterilize surgical instruments in hospitals.
Radioactive elements can also be used as tracers in medicine where they determine the efficiency of organisms such as kidneys and thyroid glands.
3. Biology and agriculture
– radioactive sources are used to generate different species of plants with new characteristics that can withstand diseases and drought. Insects are sterilized through radiation to prevent the spread of pests and diseases. Potatoes exposed to radiation can be stored for a long time without perishing.
4. Industry – thickness of metal sheets is measured accurately using radiation from radioactive sources. Recently the manufacture of industrial diamonds is undertaken through transmutation.
5. Energy source – in N. America, Europe and Russia nuclear reactors are used to generate electricity.
The amount of fuel used is quite small hence an economical way of generating electricity energy as compared to H.E.P generation.
Chapter Eleven
Electronics
Conductors, insulators and semi-conductors
i) An insulator is a material or object which resists flow of heat (thermal insulator) or electrical charges (electrical insulators). Examples are paraffin, wood, rubber, plastics etc.
ii) Conductors are materials that contain free electrons which carry an electrical charge from one point to another.
Examples are metals and non-metals like carbon, graphite etc.
iii) Semi-conductors are materials or objects which allow the flow of electrical heat or energy through them under certain conditions i.e. temperature. Examples are germanium, silicon, cadmium sulphide, gallium arsenide etc.
Electronic bond structure
This is the series of “allowed” and “forbidden” energy bands that it y bands that it contains according to the band theory which postulates the existence of continuous ranges of energy values (bands) which electron may occupy “allowed” or not occupy ‘forbidden”.
According to molecular orbital theory, if several atoms are brought together in a molecule, their atomic orbitals split, producing a number of molecular orbitals proportional to the number of atoms.
However when a large number of atoms are brought together the difference between their energy levels become very small, such that some intervals of energy contain no orbitals and this theory makes an assumption that these energy levels are as numerous as to be indistinct.
Number, size and spacing of bands.
Any solid has a large number of bands (theoretically infinite). Bands have different widths based upon the properties of the atomic orbitals from which they arise. Bands may also overlap to produce a bigger single band.
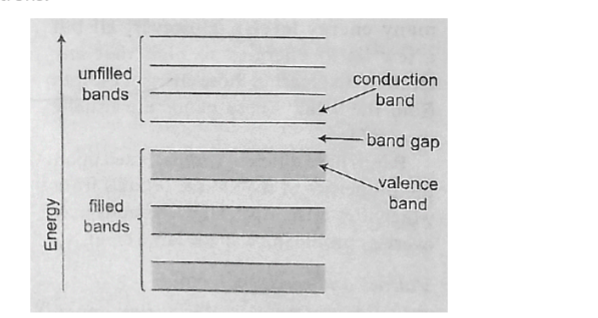
Valence and conduction bands
Valence band is the highest range of electron energies where electrons are normally present at zero temperature.
Conduction band is the range of electron energy higher than that of the valence band sufficient to make electrons free (delocalized); responsible for transfer of electric charge. Insulators and semi-conductors have a gap above valence band followed by conduction band above it. In metals, the conduction band is the valence band.
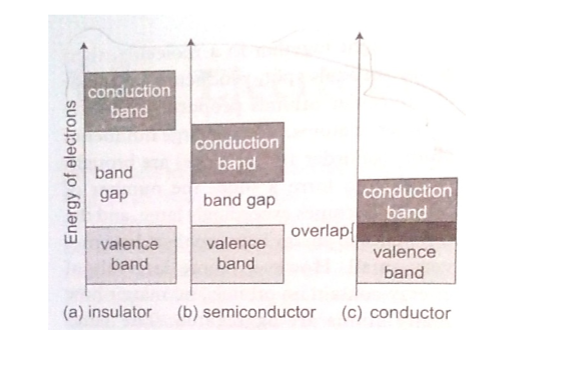
Electrons in the conduction band break free of the covalent bonds between atoms and are free to move around hence conduct charge.
The covalent bonds have missing electrons or ‘holes’ after the electrons have moved.
The current carrying electrons in the conduction band are known as free electrons.
Doping is the introduction of impurities in semi-conductors to alter their electronic properties.
The impurities are called dopants. Doping heavily may increase their conductivity by a factor greater than a million.
Intrinsic and extrinsic semi-conductors
An intrinsic semi-conductor is one which is pure enough such that the impurities in it do not significantly affect its electrical behavior.
Intrinsic semi-conductors increase their conductivity with increase in temperature unlike metals.
An extrinsic semi-conductor is one which has been doped with impurities to modify its number and type of free charge carriers present.
N-type semi-conductors
In this case the semi-conductor is given atoms by an impurity and this impurity is known as donor so it is given donor atoms (donated).
The impurity within the semi-conductor accepts atoms with free electrons (dopants). This forms a ‘hole’ within the semi-conductors.
Junction refers the region where the two types of semi-conductors meet. The junctions are made by combining an n-type and p-type semi-conductor. The n-region is the cathode and the p-region is the anode.
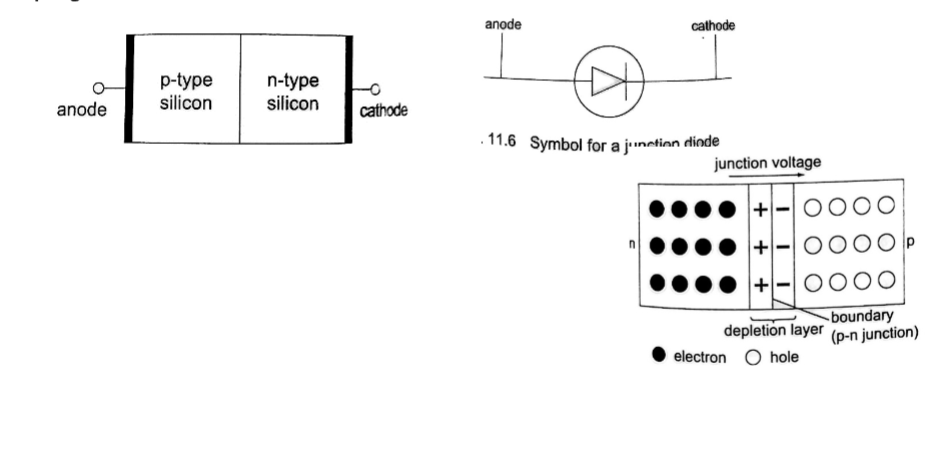
It occurs when the p-type block is connected to the positive terminal and the n-type block is connected to the negative terminal of a battery.
The depletion layer of the junction reduces to be very thin to allow the flow of electric current.
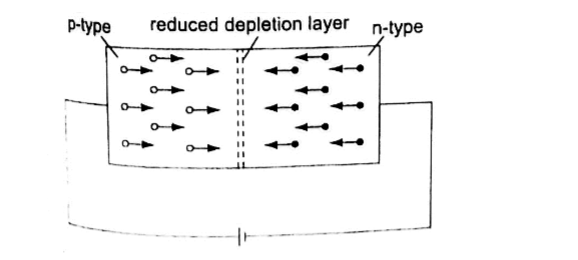
The negative terminal of the battery is connected to the p-type region while the n-type isconnected to positive terminal.
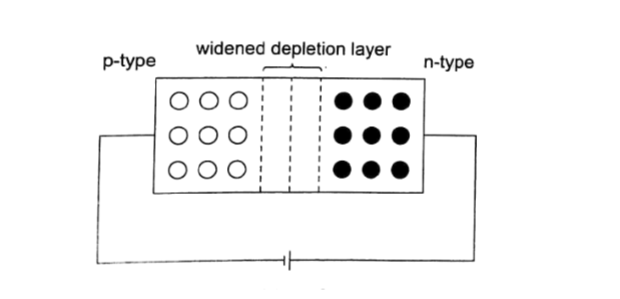
Applications of junction diodes
They are mainly used for rectification of a.c. current for use by many electrical appliances. Rectification is the conversion of sinusoidal waveform into unidirectional (non-zero) waveform.
Half wave rectification
In this case the first half cycle of a sinusoidal waveform is positive and the inclusion of a reverse biased diode makes the current not to flow to the negative side of the wave.
The current therefore conducts on every half cycle hence a half wave rectification is achieved.
The voltage is d.c. and always positive in value though it is not steady and needs to be smoothed by placing a large capacitor in parallel to the load as shown.
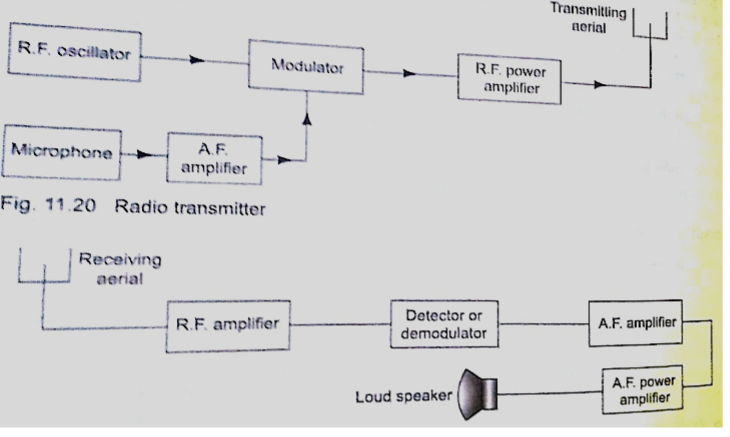
Radio waves are produced by circuits that make electrons vibrate and they are known as oscillators which produce varied frequencies. Since radio waves have greater range in air than sound or even light waves they are used as carriers of audio (sound) and visual information (TV) waves.
The sound is first changed into electrical vibrations by use of a microphone or other device then added to the radio carrier wave and this changes the amplitude of the carrier and is called amplitude modulation.
The modulated wave is given out by the transmitting aerial and received by another aerial in a radio or TV when they cause vibrations between the earth and the aerial.
They are then demodulated by a diode and hence heard as a sound or image.
KCSE Revision Notes Form 1 - Form 4 All Subjects
Secondary School Scholarships in Kenya » Kenya Postgraduate Scholarships » Undergraduate Scholarships for Kenyan Students » Kenya Scholarships for Kenyan Students Studying in Kenya » Kenya Undergraduate Scholarships » The Kenya Youth Education Scholarship Fund - Scholarships Kenya - Scholarships KCSE Results » KCSE Results Top 100 Schools - Kenya Certificate of Secondary Education – KCSE » KCSE Top 100 Candidates » Kenya Certificate of Secondary Education – KCSE » KNEC - Kenya National Examinations Council » Secondary Schools in Kenya » KNEC - Kenya National Examinations Council » Free KNEC KCSE Past Papers
Kenya Scholarships for Undergraduate Students » Kenya Scholarships for Postgraduate Students » Undergraduate Scholarships for Kenyan Students » Kenya Undergraduate Scholarships » Full Undergraduate Scholarships for Kenyans » Kenya Postgraduate Scholarships » Scholarships & Grants » Undergraduate Scholarships » Universities in Kenya » Kenya Universities and Colleges Central Placement Service (KUCCPS) » Colleges in Kenya » KASNEB Registration & Results » Secondary Schools Scholarships in Kenya » Undergraduate & Graduate Scholarships for Kenyans
Powerful Motivational Quotes for Students » Success Quotes for Students » KCSE Motivational Quotes for KCSE Candidates » KCSE Success Quotes for KCSE Candidates
a a a physics notes! chapter 1 introduction to physics college physics notes download klb physics book 4 download physics notes form 3 electronics form four notes form 1 past papers form 1 past papers with answers form 1 physics notes form 1 physics questions and answers form 1 physics revision notes form 1 physics syllabus form 1 physics test paper pdf form 2 physics exam paper form 2 physics exam paper 2016 form 2 physics exam paper free download form 2 physics exam paper with answer form 2 physics final year exam paper 2 form 2 physics past papers form 2 physics revision notes form 2 physics short notes form 2 physics syllabus form 3 past papers form 3 physics exam paper form 3 physics notes form 3 physics past papers form 3 physics questions form 3 physics questions and answers pdf form 3 physics revision notes form 3 physics syllabus form 4 exam papers form 4 physics notes form 4 physics revision notes form 4 physics syllabus form 4 physics topics form 5 physics topics form five physics notes form four physics notes form four physics questions and answers form four physics questions and answers pdf form four physics topics form four revision papers form one exams form one past papers form one physics examination form one physics past papers pdf form one physics questions and answers form one physics questions and answers pdf form one physics topics form one term one physics exam form three physics notes form three physics notes pdf form three physics questions and answers form three physics questions and answers pdf form three physics topics form two notes form two physics notes form two physics notes pdf form two physics questions and answers form two physics questions and answers pdf form two physics syllabus form two physics topics high school physics notes high school physics study guide introduction of physics form one introduction to physics notes k.l.b physics notes kcse physics notes kcse physics syllabus kenya secondary school physics syllabus pdf klb physics book 1 pdf klb physics book 2 pdf klb physics book 3 pdf klb physics book 3 pdf download klb physics book 4 notes klb physics book 4 pdf klb physics book 4 pdf download klb physics book 4 topics klb physics form 1 klb physics form 1 pdf klb physics form 2 klb physics form 2 notes klb physics form 2 pdf klb physics form 2 pdf download klb physics form 3 klb physics form 3 pdf klb physics form 3 pdf download klb physics form 4 klb physics form 4 pdf klb physics form four notes klb physics form one notes klb physics form three notes klb physics form two notes klb physics notes klb physics notes form 4 klb physics pdf maktaba tetea notes necta form four past papers necta past papers form 4 2016 necta past papers form six necta past papers form two necta physics past papers necta physics practicals necta questions and answers necta review questions notes za physics form one notes za physics form three past papers 2014 physic form 4 chapter 1 mind map physic notes physics book 4 pdf physics exam form three physics form 1 exams physics form 1 mid year exam physics form 1 past papers physics form 1 pressure physics form 1 questions and answers physics form 1 questions and answers pdf physics form 2 exam paper 2014 physics form 2 exams physics form 2 notes physics form 2 past papers physics form 2 pdf physics form 2 questions and answers physics form 2 questions and answers pdf physics form 3 exams physics form 3 notes pdf physics form 3 past papers physics form 3 questions and answers physics form 3 syllabus physics form 4 chapter 1 conversion of units physics form 4 chapter 1 exercise physics form 4 chapter 1 exercise and answers physics form 4 chapter 1 exercise pdf physics form 4 chapter 1 mind map physics form 4 chapter 2 physics form 4 chapter 2 exercise and answers physics form 4 chapter 2 exercise pdf physics form 4 chapter 2 experiment physics form 4 chapter 2 formula physics form 4 chapter 2 mind map physics form 4 chapter 2 momentum physics form 4 chapter 2 notes pdf physics form 4 chapter 2 objective questions and answers physics form 4 chapter 2 paper 2 physics form 4 chapter 2 slideshare physics form 4 chapter 3 physics form 4 chapter 3 questions and answers physics form 4 chapter 4 notes pdf physics form 4 chapter 5 light questions and answers physics form 4 chapter 5 notes pdf physics form 4 exam paper 1 physics form 4 exams physics form 4 exercise physics form 4 exercise pdf physics form 4 module with answer physics form 4 notes chapter 1 physics form 4 notes free download physics form 4 notes pdf physics form 4 paper 2 questions and answers physics form 4 past papers physics form 4 questions and answers physics form 4 revision notes physics form 5 chapter 1 exercise and answers physics form 5 chapter 1 notes pdf physics form 5 chapter 2 notes pdf physics form 5 chapter 2 slideshare physics form 5 chapter 3 notes pdf physics form 5 notes pdf physics form four book physics form four notes pdf physics form four questions physics form four study notes physics form four topics physics form one physics form one book physics form one notes physics form one notes pdf physics form one study notes physics form three book physics form three notes physics form three study notes physics form two book physics form two notes physics form two notes pdf physics form two questions physics form two study notes physics form two topics physics module form 5 physics notes physics notes for class 11 pdf physics notes for class 12 pdf physics notes form 1 free download physics notes igcse physics notes pdf physics simple notes physics spm notes download physics spm notes pdf physics spm questions physics study guide answers physics study guide pdf physics study guides radioactivity form four secondary physics notes pdf spm notes success physics spm pdf tahossa past papers
1 a a kcse past papers 2014 kcse marking schemes 2016 kcse papers 2016 kcse prediction questions 2018 kcse exam 2018 kcse questions a a kcse past papers advance-africa.com kcse rev quiz agriculture mock papers agriculture paper 2 questions and answers pdf alliance mocks 2017 ap biology essay questions and answers arabic exam 2016 arabic oral exam questions betrayal in the city essay questions and answers pdf betrayal in the city essay questions with answers betrayal in the city, ,,revision questions biology book 3 klb biology essay questions and answers form 4 biology essay questions and answers form 4 pdf biology essays pdf biology exam questions and answers pdf biology form 2 questions and answers pdf biology form 3 notes pdf biology form 3 questions and answers pdf biology form 3 syllabus biology form three reproduction biology form three-questions and answers biology kcse - kcse biology questions and answers - kcse biology essay questions and answers - kcse biology paper 1 2015 - kcse biology notes - kcse 2015 biology paper 2 - kcse biology practical 2015 - kcse biology practicals - kcse biology 2011
biology kcse 2017 biology kcse questions biology paper 1 questions and answers biology paper 2 questions and answers biology paper 3 questions and answers biology questions and answers for high schools biology questions and answers for high schools pdf biology questions and answers form 2 biology questions and answers multiple choice biology questions and answers on cells biology questions and answers online biology questions and answers pdf biology revision notes form 3 business past kcse past papers c.r.e form one notes pdf cambridge igcse computer science cambridge igcse computer science answers cambridge igcse computer science coursebook pdf download cambridge igcse computer science revision guide pdf cambridge igcse computer science study and revision guide pdf cambridge igcse computer science workbook - free download cambridge igcse computer science workbook pdf caucasian chalk circle essay questions chemistry paper 1 questions and answers chemistry paper 2 questions and answers chemistry paper 3 question and answer chemistry past papers form 1 chemistry past papers form 2 cie past papers computer science 0478 computer science igcse past papers xtremepapers computer science paper 2 2017 computer science past papers a level computer science past papers o level computer studies form 1 questions computer studies form 3 past papers computer studies past papers computer studies questions and answers pdf county mocks 2017 cre form 2 notes pdf cre form 3 notes cre form 3 notes pdf cre form 4 notes cre form 4 notes pdf cre form one notes cre kcse 2016 cre notes cre notes form 2 cre notes pdf cre paper 1 with answers cre paper 2 cre paper 2 topics cre preparation notes cre questions form one cre revision notes cre revision questions and answers download kcse past papers with answers dvance kcse past papers edexcel igcse computer science past papers english paper 3 question paper - 2014 kcse english paper 3 question paper - 2015 kcse english paper 3 question paper - 2016 kcse english paper 3 question paper - 2017 kcse english paper 3 question paper - 2018 kcse essay questions and answers on betrayal in the city essay questions based on betrayal in the city find download kcse past papers with answers - kcse past papers pdf download - kcse 2013 marking scheme - kcse mathematics past papers pdf - free kcse past papers and marking schemes - kcse mock papers pdf - kcse past papers 2014 pdf - kcse past papers 2015 - kcse past papers 2010 find kcse biology essay questions and answers - kcse biology practicals - kcse biology paper 1 2015 - biology essay questions and answers form 4 - kcse biology questions and answers - ap biology essay questions and answers - kcse biology notes - kcse biology paper 2 2012 - kcse biology paper 2 2015
form 2 biology questions and answers free kcse mocks 2015 free kcse past papers - kcse past papers - knec kcse online past papers - knec kcse results past papers free kcse past papers 2014 free kcse past papers kenya, free marking schemes, download ... free kcse past papers with answers free kcse questions and answers on chemistry free revision papers general biology test questions and answers general science questions and answers pdf history and government paper one topics history form one questions and answers pdf history paper 1 questions and answers history paper 2 questions and answers home science past papers igcse computer science book igcse computer science book pdf download igcse computer science notes igcse computer science paper 2 notes igcse computer science past papers igcse computer science past papers 2014 igcse computer science past papers 2017 igcse computer science pdf igcse computer science pre release material 2018 igcse computer science resources igcse computer science revision notes pdf igcse computer science workbook pdf igcse computer studies past papers interesting biology questions ire kcse past papers k.c.s.e cre paper 1 2017 k.c.s.e geography 2017 k.c.s.e mathematics paper 1 2017 k.c.s.e mocks 2018 k.c.s.e past papers 2014 kcpe 2018 predictions kcpe prediction questions kcse 2010 marking scheme kcse 2010 past papers kcse 2011 cre paper 1 kcse 2011 marking scheme kcse 2012 history paper 2 marking scheme kcse 2012 marking schemes kcse 2013 cre paper 1 kcse 2013 marking scheme kcse 2013 marking scheme pdf kcse 2014 kcse 2015 biology paper 2 kcse 2015 biology paper 3 kcse 2015 marking scheme kcse 2015 past papers kcse 2016 agriculture paper 2 kcse 2016 biology paper 1 kcse 2016 biology paper 2 kcse 2016 computer paper 1 kcse 2017 marking scheme kcse 2017 maths paper 1 kcse 2017 papers kcse 2017 papers and marking scheme kcse 2017 past papers kcse 2017 prediction pdf kcse 2018 cre prediction kcse 2018 leakage kcse 2018 marking scheme kcse 2018 papers kcse 2018 predictions kcse 2019 marking scheme kcse agriculture past papers kcse answers kcse arabic paper 1 kcse arabic paper 2 kcse arabic paper 3 kcse arabic paper 3 2016 kcse arabic past papers kcse biology 2011 kcse biology essay questions and answers kcse biology essay questions and answers - kcse revision questions and answers - kcse chemistry questions and answers - kcse revision papers with answers - kcse past papers with answers - download kcse past papers with answers - kcse questions on the river and the source - kcse revision notes
kcse biology essay questions and answers - kcse revision questions and answers - kcse chemistry questions and answers - kcse revision papers with answers - kcse past papers with answers - download kcse past papers with answers - kcse questions on the river and the source - kcse revision notes
kcse biology essay questions and answers pdf kcse biology essays kcse biology essays pdf kcse biology notes kcse biology paper 1 kcse biology paper 1 2017 kcse biology paper 1 2017 pdf kcse biology paper 2 2012 kcse biology paper 2 2015 kcse biology paper 2 2017 kcse biology paper 3 2016 kcse biology paper 3 past papers kcse biology past papers kcse biology past papers and answers kcse biology practical 2016 kcse biology practical past papers kcse biology practicals kcse biology questions and answers kcse biology questions and answers - kcse past papers biology - kcse biology essay questions and answers - kcse chemistry past papers - download kcse past papers with answers - k.c.s.e papers 2015 - k.c.s.e papers 2016 - kcse biology paper 1 2015 - kcse past papers 2015 - kcse past papers 2011 - kcse past papers 2016 - kcse past papers 2017 - 2017 kcse prediction questions - 2018 kcse prediction questions
kcse business paper 1 2016 kcse business past papers kcse business studies past papers kcse chemistry paper 1 2016 kcse chemistry paper 1 2017 kcse chemistry paper 3 2012 kcse chemistry past papers kcse chemistry past papers and answers kcse chemistry practical kcse computer studies paper 1 kcse computer studies paper 2 kcse computer studies paper 2 pdf kcse cre 2016 kcse cre paper 1 2013 kcse cre paper 1 2015 kcse cre paper 1 2016 kcse cre paper 1 2017 kcse cre paper 2 kcse cre paper 2 2016 kcse cre past papers kcse cre past papers and answers kcse english paper 3 2016 kcse english paper 3 2017 kcse essay questions in betrayal in the city kcse exam papers 2018 kcse exam papers answers kcse french paper 1 kcse french paper 2 kcse french past papers kcse general science syllabus kcse geography paper 2 2016 kcse history paper 1 2012 kcse history paper 2 2016 kcse history paper 2 2017 kcse kiswahili paper 1 2017 kcse marking scheme 2016 kcse marking schemes kcse marking schemes 2017 kcse marking schemes pdf kcse mathematics marking schemes kcse mathematics paper 1 2015 kcse mathematics paper 1 2016 kcse mathematics paper 2 2016 kcse mathematics past papers kcse mathematics past papers pdf kcse mock exams kcse mock papers 2015 kcse mock papers 2017 kcse mock papers 2018 kcse mock papers pdf kcse mock papers pdf 2018 kcse mocks 2017 kcse mocks 2018 kcse music past papers kcse online past papers kcse papers 2015 kcse past papers kcse past papers - kcpe and answers - free mocks online - kcse answers past exams question papers - downloads | kcse papers and marking schemes | exams - kcse mathematics paper 1 questions and answers - kcse cre paper 1 questions and answers - knec past papers free downloads - kcse online registration - kcpe - kcse past papers - knec - knec portal - knec past papers for colleges - kasneb - past papers - kasneb past papers for colleges - cpa past papers - https://www.knec.ac.ke/ - www.knec-portal.ac.ke/ - knec portal: kcse results, online registration, kcse result slip. knec portal confirmation - knec portal kcse results - knec examiners portal - knec website kcse past papers - kcpe and answers - free mocks online - kcse answers past exams question papers - downloads | kcse papers and marking schemes | exams - kcse mathematics paper 1 questions and answers - kcse cre paper 1 questions and answers
kcse past papers 2007 kcse past papers 2009 kcse past papers 2010 kcse past papers 2011 kcse past papers 2011 pdf kcse past papers 2012 kcse past papers 2013 kcse past papers 2013 -knec kcse past papers 2014 kcse past papers 2014 pdf kcse past papers 2015 kcse past papers 2015 marking schemes kcse past papers 2015 pdf kcse past papers 2016 kcse past papers 2016 pdf kcse past papers 2017 kcse past papers 2017 pdf kcse past papers agriculture and answers kcse past papers arabic and answers kcse past papers art and design and answers kcse past papers biology kcse past papers building and construction and answers kcse past papers business studies and answers kcse past papers chemistry kcse past papers chemistry and answers kcse past papers chemistry pdf kcse past papers computer studies and answers kcse past papers cre and answers kcse past papers electricity and answers kcse past papers english and answers kcse past papers french and answers kcse past papers general science and answers kcse past papers geography and answers kcse past papers german and answers kcse past papers history and government and answers kcse past papers home science and answers kcse past papers hre and answers kcse past papers ire and answers kcse past papers kenya sign language and answers kcse past papers kiswahili and answers kcse past papers marking scheme kcse past papers maths kcse past papers metal work and answers kcse past papers music and answers kcse past papers pdf download kcse past papers physics and answers kcse past papers physics with answers kcse past papers power mechanics and answers kcse past papers with answers kcse past papers woodwork and answers kcse physics past papers kcse prediction 2017 kcse prediction 2018 kcse prediction 2018 pdf kcse prediction papers 2018 kcse prediction questions 2018 kcse prediction questions and answers kcse questions and answers kcse questions and answers. download free kcse past papers from knec. all marking schemes - questions and answers are sourced from knec. kcse revision kcse revision papers 2014 kcse revision | secondary school | text books | text book centre kcse trial 2017 kcse trial exams 2017 kenyaplex kcse past papers kenyaplex past papers for secondary kiswahili paper 3 questions and answers klb biology form 3 pdf klb cre form 1 klb cre form 3 knec ict past papers knec past papers for colleges knec past papers free download knec past papers pdf knec revision papers knec technical exams past papers kusoma.com past papers maths kcse 2017 mock past papers 2017 mock past papers with answers mokasa mock 2017 page navigation papacambridge computer science igcse past kcse papers past papers in kenya pre mocks 2018 pte knec past papers revision sample essays on betrayal in the city school biology notes school geography notes school physics notes school river and the source themes used in betrayal in the city xtremepapers igcse computer science z notes computer science igcse
Physics Notes Form 4 - Physics Notes Form Four - Notes za Physics Notes Form 4
"Pdf" Revision Questions Physics Form 1 "Pdf" Revision Questions Physics Form 2 "Pdf" Revision Questions Physics Form 3 "Pdf" Revision Questions Physics Form 4 "Pdf" Revision Questions Physics Form Four "Pdf" Revision Questions Physics Form One "Pdf" Revision Questions Physics Form Three "Pdf" Revision Questions Physics Form Two 1 a a KCSE Past Papers 10th Grade Physics Questions and Answers 10th Grade Physics Test 11th Ncert Physics 12th Class Physics Book Free Download 2014 KCSE Marking Schemes 2014 Pdf KCSE Past Papers 2015 2015 Physics Essay Questions and Answers Form 4 2016 KCSE Papers 2016 KCSE Prediction Questions 2017 Physics Hsc Answers 2017 KCSE Prediction Questions 2018 Physics KCSE Leakage 2018 Physics KCSE Questions 2018 KCSE Busineness Studies 2018 KCSE Exam 2018 KCSE Leakage 2018 KCSE Prediction Questions 2018 KCSE Questions 2019 Physics KCSE Leakage 2019 Physics KCSE Questions 2019 KCSE Leakage 2019 KCSE Questions 9th Grade Physics Study Guide A a a Physics Notes a a a Physics Notes! a a a PhysicsNotes! A a KCSE Past Papers A Biblical View of Social Justice A Level Physics Biological Molecules Questions A Level Physics Exam Questions by Topic A Level Physics Notes Edexcel A Level Physics Notes Xtremepapers A Level Physics Past Papers A Level Physics Questions and Answers a Level Physics Questions and Answers A Level Physics Questions and Answers (Pdf) A Level Physics Questions and Answers Pdf A Level Physics Questions by Topic Kidney Questions With Markschemes A Level Physics Revision A Level Physics Revision Edexcel A Level Physics Revision Guide A Level Physics Revision Notes A Level Physics Revision Notes Pdf A Level Physics Textbook Pdf A Level Physics Year 1 / as Aqa Exam Questions by Topic A Level Edexcel Notes a* Physics aa Physics Form 3 Questions and Answers Advance KCSE Past Papers Advance-africa.com KCSE Rev Quiz Advantages and Disadvantages. All Physics Essays All Physics Notes for Senior Two All KCSE Past Papers Physics With Making Schemes All Marking Schemes Questions and Answers All Past K.c.s.e Questions With Answers Alliance Mocks 2017 Ap Bio Quizzes Ap Physics 1 Textbook Pdf Ap Physics Essay Questions and Answers Are Sourced From KNEC. As Level Physics Notes Atika Physics Notes Atika School Physics Notes B/s Book 2 Notes Basic Physics Books Pdf basic Physics Interview Questions and Answers Pdf Basic Physics Interview Questions and Answers Pdf Basic Physics Pdf Basic Physics Questions and Answers Basic Physics Questions and Answers Pdf Bbc Bitesize Physics Ks3 Bihar Board Physics Objective Answer 2017 Bihar Board Physics Objective Answer 2018 Bio Answers Bio Quesions Physics 0478 Physics 101 Physics 12th Physics 12th Class Notes Pdf Physics 2019 Syllabus Physics All KCSE Short Notes Physics Answers Physics Answers Online Free Physics Answers Quizlet Physics Bk 2 Notes Physics Book 1 Physics Book 1 Notes Physics Book 2 Physics Book 2 Notes Physics Book 3 Physics Book 3 KLB Physics Book 3 Notes Physics Book 4 Physics Book 4 Notes Physics Book 4 Pdf Physics Book for Class 11 Physics Book Four Physics Book Four Notes Physics Book One Physics Book One Notes Physics Book Pdf Free Download Physics Book Three Physics Book Three Notes Physics Book Three Pdf Physics Book Two Physics Book Two Notes Physics Books Form Three Physics Bowl Physics Study Guide Physics Bowl Questions Physics Physics Bowl Questions Earth Physics Physics Bowl Questions Math Physics Bowl Questions Middle School Physics Brekthrough Form Two Notes Physics Class 12 Ncert Solutions Physics Class 12 Pdf Physics Communication Syllabus Physics Diagram Software Physics Diagrams for Class 11 Physics Diagrams for Class 12 Physics Diagrams for Class 9 Physics Diagrams for Class-10 Physics Diagrams in Form 1 Physics Diagrams in Form 2 Physics Diagrams in Form 3 Physics Diagrams in Form 4 Physics Diagrams Pdf Physics Diagrams to Label Physics Essay Questions and Answers Physics Essay Questions and Answers 2018 Physics Essay Questions and Answers Form 1 Physics Essay Questions and Answers Form 2 Physics Essay Questions and Answers Form 3 Physics Essay Questions and Answers Form 4 Physics Essay Questions and Answers Form 4 Pdf Physics Essay Questions and Answers Pdf Physics Essay Revision Q Physics Essays and Answers Physics Essays Form One to Form Four Physics Essays Form One to Form Three Physics Essays KCSE Physics Essays Pdf Physics Exam 1 Multiple Choice Physics Exam 2 Advance Physics Exam 2 Test Physics Exam 2016 Physics Exam Form Four Physics Exam Form One Physics Exam Form Three Physics Exam Form Two Physics Exam Practice Test Physics Exam Questions Physics Exam Questions and Answers Physics Exam Questions and Answers Pdf Physics Exam Study Guide Physics Exams Physics Excretion Notes Physics Exercise Form 4 With Answers Physics Final Exam Answer Key Physics Final Exam Answer Key 2016 Physics Final Exam Answer Key 2017 Physics Final Exam Answers 2018 Physics Final Exam Answers 2019 Physics Final Exam Questions and Answers Physics Fom 1 Notes Physics Fom 2 Notes Physics Fom 3 Notes Physics Fom 4 Notes Physics Form 1 Physics Form 1 & 2 and Answers Physics Form 1 and 2 Essays Physics Form 1 and 2 Essays Questions and Answers Physics Form 1 Chapter 1 Physics Form 1 Diagrams Physics Form 1 Exams Physics Form 1 Mid Year Exam Physics Form 1 Notes Physics Form 1 Notes and Questions Physics Form 1 Notes Download Physics Form 1 Notes Free Download Physics Form 1 Notes GCSE Physics Form 1 Notes KCSE-kcse Physics Form 1 Notes Pdf Physics Form 1 Notes Pdf Download Physics Form 1 Past Papers Physics Form 1 Pdf Physics Form 1 Pressure Physics Form 1 Question Papers Physics Form 1 Questions Physics Form 1 Questions and Answers Physics Form 1 Questions and Answers Pdf Physics Form 1 Quiz Physics Form 1 Revision Questions Physics Form 1 Summary Notes Physics Form 1 Syllabus Physics Form 1 Work Physics Form 1-4 Notes Physics Form 2 Physics Form 2 Chapter 1 Physics Form 2 Chapter 2 Physics Form 2 Diagrams Physics Form 2 Exam Paper 2014 Physics Form 2 Exams Physics Form 2 Notes Physics Form 2 Notes and Questions Physics Form 2 Notes GCSE Physics Form 2 Notes KCSE-kcse Physics Form 2 Notes Pdf Physics Form 2 Notes Pdf Download Physics Form 2 Past Papers Physics Form 2 Pdf Physics Form 2 Question Papers Physics Form 2 Questions Physics Form 2 Questions and Answers Physics Form 2 Questions and Answers Pdf Physics Form 2 Quiz Physics Form 2 Revision Notes Physics Form 2 Salts Physics Form 2 Structure and Bonding Physics Form 2 Summary Notes Physics Form 2 Syllabus Physics Form 2 Work Physics Form 3 Physics Form 3 and 4 Essays Physics Form 3 and 4 Essays Questions and Answers Physics Form 3 Chapter 3 Physics Form 3 Classification Physics Form 3 Diagrams Physics Form 3 Ecology Physics Form 3 Exams Physics Form 3 Notes Physics Form 3 Notes and Questions Physics Form 3 Notes GCSE Physics Form 3 Notes KCSE-kcse Physics Form 3 Notes Pdf Physics Form 3 Notes Pdf Download Physics Form 3 Notes Topic 1 Physics Form 3 Past Papers Physics Form 3 Pdf Physics Form 3 Question Papers Physics Form 3 Questions Physics Form 3 Questions and Answers Physics Form 3 Questions and Answers Pdf Physics Form 3 Questions and Answers Term 3 Physics Form 3 Questions and Answers+pdf Physics Form 3 Quiz Physics Form 3 Revision Notes Physics Form 3 Revision Questions Physics Form 3 Summary Notes Physics Form 3 Syllabus Physics Form 3 Syllabus Pdf Physics Form 3 Topics Physics Form 3 Work Physics Form 4 Physics Form 4 All Chapter Physics Form 4 Chapter 1 Conversion of Units Physics Form 4 Chapter 1 Exercise Physics Form 4 Chapter 1 Exercise and Answers Physics Form 4 Chapter 1 Exercise Pdf Physics Form 4 Chapter 1 Mind Map Physics Form 4 Chapter 2 Physics Form 4 Chapter 2 Exercise and Answers Physics Form 4 Chapter 2 Exercise Pdf Physics Form 4 Chapter 2 Experiment Physics Form 4 Chapter 2 Formula Physics Form 4 Chapter 2 Mind Map Physics Form 4 Chapter 2 Momentum Physics Form 4 Chapter 2 Notes Pdf Physics Form 4 Chapter 2 Objective Questions and Answers Physics Form 4 Chapter 2 Paper 2 Physics Form 4 Chapter 2 Slideshare Physics Form 4 Chapter 3 Physics Form 4 Chapter 3 Questions and Answers Physics Form 4 Chapter 4 Physics Form 4 Chapter 4 Notes Pdf Physics Form 4 Chapter 5 Light Questions and Answers Physics Form 4 Chapter 5 Notes Pdf Physics Form 4 Diagrams Physics Form 4 Exam Paper 1 Physics Form 4 Exams Physics Form 4 Exercise Physics Form 4 Exercise Pdf Physics Form 4 Module With Answer Physics Form 4 Note Physics Form 4 Notes Physics Form 4 Notes (Pdf) Physics Form 4 Notes All Chapter Pdf Physics Form 4 Notes and Questions Physics Form 4 Notes Chapter 1 Physics Form 4 Notes Chapter 2 Physics Form 4 Notes Chapter 3 Physics Form 4 Notes Download Physics Form 4 Notes Free Download Physics Form 4 Notes GCSE Physics Form 4 Notes KCSE-kcse Physics Form 4 Notes Pdf Physics Form 4 Notes Pdf Download Physics Form 4 Paper 2 Questions and Answers Physics Form 4 Past Papers Physics Form 4 Question Papers Physics Form 4 Questions Physics Form 4 Questions and Answers Physics Form 4 Questions and Answers Pdf Physics Form 4 Quiz Physics Form 4 Revision Notes Physics Form 4 Schemes of Work Physics Form 4 Summary Notes Physics Form 4 Syllabus Physics Form 4 Textbook Pdf Physics Form 4 Work Physics Form 5 Chapter 1 Exercise and Answers Physics Form 5 Chapter 1 Notes Pdf Physics Form 5 Chapter 2 Notes Pdf Physics Form 5 Chapter 2 Slideshare Physics Form 5 Chapter 3 Notes Pdf Physics Form 5 Notes Pdf Physics Form Four Book Physics Form Four Notes Physics Form Four Notes and Questions Physics Form Four Notes GCSE Physics Form Four Notes Pdf Physics Form Four Past Papers Physics Form Four Questions Physics Form Four Questions and Answers Physics Form Four Questions and Answers Pdf Physics Form Four Quiz Physics Form Four Study Notes Physics Form Four Syllabus Physics Form Four Topic 2 Physics Form Four Topic 4 Physics Form Four Topics Physics Form Four Work Physics Form One Physics Form One Book Physics Form One Book Pdf Physics Form One Download Topic 1 Upto 3 Physics Form One Exam Physics Form One Notes Physics Form One Notes and Questions Physics Form One Notes GCSE Physics Form One Notes Pdf Physics Form One Pdf Physics Form One Questions Physics Form One Questions and Answers Physics Form One Questions and Answers Pdf Physics Form One Questions and Their Answers Physics Form One Quiz Physics Form One Revision Question Physics Form One Schemes of Work Physics Form One Study Notes Physics Form One Syllabus Physics Form One Term Three Test Physics Form One to Three Notes Physics Form One Work Physics Form Three Physics Form Three Book Physics Form Three Notes Physics Form Three Notes and Questions Physics Form Three Notes GCSE Physics Form Three Questions and Answers Physics Form Three Questions and Answers Pdf Physics Form Three Quiz Physics Form Three Reproduction Physics Form Three Reproduction. Physics Form Three Study Notes Physics Form Three Work Physics Form Three-questions and Answers Physics Form Two Physics Form Two Book Physics Form Two Diagrams Physics Form Two Notes Physics Form Two Notes and Questions Physics Form Two Notes GCSE Physics Form Two Notes Pdf Physics Form Two Notes-pdf Physics Form Two Pdf Physics Form Two Questions Physics Form Two Questions and Answers Physics Form Two Questions and Answers Pdf Physics Form Two Quiz Physics Form Two Study Notes Physics Form Two Topics Physics Form Two Work Physics Form Two,schemes of Work Physics Form2 Physics Form2 Textbook Physics Game Form Four Question End Answers Physics Grade 10 Exam Papers Physics Hsc Pdf Physics Human Reproduction Video Physics IGCSE Past Papers Xtremepapers Physics K.c.s.e 2017 Physics KCSE Physics KCSE 2016 Physics KCSE 2017 Physics KCSE 2017 Paper 1 Physics KCSE Past Papers Physics KCSE Questions Physics KCSE Questions and Answer Physics KCSE Quizzes & Answers Physics KCSE Revision Physics KCSE Revision Notes Physics KCSE Setting Questions Form One and Two Physics Ksce 2015 Physics Last Year K.c.s.e Questions Physics Lesson Plan Form Two Physics Made Familiar Physics Mcq for Class 11 Physics Mcq for Class 12 Physics Mcq for Competitive Exams Physics Mcq for Competitive Exams Pdf Physics Mcq for Neet Pdf Physics Mcq for Ssc Physics Mcq Questions With Answers Physics Mcq With Answers Pdf Physics Mcqs for Class 12 Pdf Physics Mcqs With Answers Pdf Physics Mid Familia Form One Physics Mock Papers Physics Module Form 5 Physics Multiple Choice Questions and Answers Cxc Physics Multiple Choice Questions and Answers Pdf Physics Multiple Choice Questions With Answers Pdf Physics Note Physics Note Form Two All Chapters Physics Notes Physics Notes and Guestion and Answear Physics Notes and Syllabus Physics Notes Class 10 Physics Notes for Class 11 Pdf Physics Notes for Class 12 Pdf Physics Notes for High School Students Physics Notes for IGCSE 2014 Physics Notes Form 1 Physics Notes Form 1 4 Physics Notes Form 1 Free Download Physics Notes Form 1 KLB Physics Notes Form 1 Pdf Physics Notes Form 1-4 Physics Notes Form 1-4(1) Physics Physics Notes Form 14 Physics Notes Form 2 Physics Notes Form 2 KLB Physics Notes Form 2 Pdf Physics Notes Form 2; Physics Notes Physics Notes Form 3 Physics Notes Form 3 KLB Physics Notes Form 3 Pdf Physics Notes Form 4 Physics Notes Form 4 Chapter 2 Physics Notes Form 4 KLB Physics Notes Form 4 Pdf Physics Notes Form 4-pdf Physics Notes Form Four Physics Notes Form Four KLB Physics Notes Form Four Pdf Physics Notes Form One Physics Notes Form One KLB Physics Notes Form One Pdf Physics Notes Form One to Form Four Physics Notes Form Three Physics Notes Form Three KLB Physics Notes Form Three Pdf Physics Notes Form Two Physics Notes Form Two KLB Physics Notes Form Two Pdf Physics Notes Form2 Physics Notes IGCSE Physics Notes Kenya Physics Notes on Agroforestry Physics Notes Pdf Physics Notes: Physics Objective Answer Physics Objective Answer 2018 Physics Objective Questions for Competitive Exams Physics Objective Questions for Competitive Exams Pdf Physics Oral Exam Questions Physics Paper 1 Physics Paper 1 2018 Marking Rules Physics Paper 1 Notes Physics Paper 1 Questions Physics Paper 1 Questions and Answers Physics Paper 1 Topics Physics Paper 1 With Answers Physics Paper 2 Physics Paper 2 2017 Physics Paper 2 2018 Marking Rules Physics Paper 2 Questions and Answers Physics Paper 2 Questions and Answers Pdf Physics Paper 2 Revision Physics Paper 2 Topics Physics Paper 2018 Physics Paper 3 2018 Marking Rules Physics Paper 3 Question and Answer Physics Paper 3 Question Paper 2014 KCSE Physics Paper 3 Question Paper 2015 KCSE Physics Paper 3 Question Paper 2016 KCSE Physics Paper 3 Question Paper 2017 KCSE Physics Paper 3 Question Paper 2018 KCSE Physics Paper 3 Questions and Answers Physics Paper One Questions and Answers Physics Paper One Topics Physics Paper Two Qestions With Answers Physics Paper1 Physics Paper2 Physics Paper3 Physics Paper4 Physics Past Papers Physics Past Papers 2017 Physics Past Papers a Level Physics Past Papers Form 1 Physics Past Papers Form 2 Physics Past Papers Form 3 Physics Past Papers O Level Physics Pdf Download Physics Pp1 KCSE 2016 Physics Practical Book Class 12 Pdf Physics Practical Exam Physics Practicals Form One Physics Practicals Questions and Answers Physics Practice Test 9th Grade Physics Practice Test Answers Physics Practice Test Questions and Answers Physics Practice Test Quizlet Physics Predicted Questions This Year KCSE Physics Preparation Notes Physics Pretest High School Pdf Physics Question and Answer With Explanation Physics Question and Answers 2019 Physics Question and Answers 2020 Physics Question and Answers 2021 Physics Question and Answers 2022 Physics Question and Answers Note Physics Questions Physics Questions and Answers Physics Questions and Answers for High School Physics Questions and Answers for High Schools Physics Questions and Answers for High Schools Pdf Physics Questions and Answers for Secondary Schools Physics Questions and Answers Form 1 Physics Questions and Answers Form 2 Physics Questions and Answers Form 3 Physics Questions and Answers Form 4 Physics Questions and Answers Multiple Choice Physics Questions and Answers Notes Physics Questions and Answers O Physics Questions and Answers Online Physics Questions and Answers Pdf Physics Questions and Answers Pdf for Class 12 Physics Questions and Answers Pdf for Competitive Exams Physics Questions and Answers-form 2 Physics Questions for High School Physics Questions for High School Students With Answers Physics Questions for Senior 1 Physics Questions for Senior 2 Physics Questions for Senior 3 Physics Questions for Senior 4 Physics Questions for Senior 5 Physics Questions for Senior 6 Physics Questions for Senior Five Physics Questions for Senior Four Physics Questions for Senior One Physics Questions for Senior Six Physics Questions for Senior Three Physics Questions for Senior Two Physics Questions Form One Physics Questions Multiple Choice Physics Questions Quizlet Physics Questions to Ask Your Teacher Physics Quetion and Answer Form Four Physics Quetion and Answer Form One Physics Quetion and Answer Form Three Physics Quetion and Answer Form Two Physics Quiz for Class 9 Physics Quiz for Class 9 Physics Physics Quiz Questions and Answers for Class 10 Physics Quiz Questions and Answers for Class 10 Pdf Physics Quiz Questions and Answers for Class 12 Physics Quiz Questions and Answers for Class 9 Physics Quiz Questions and Answers for Class 9 Pdf Physics Quiz Questions and Answers for High School Physics Quiz Questions and Answers Multiple Choice Physics Quiz Questions and Answers Pdf Physics Quiz Questions for Class 12 Physics Quiz Questions for College Students Physics Quiz With Answers Physics Quiz With Answers Pdf Physics Quizlet Physics Revision Physics Revision a Level Physics Revision Physics Notes Physics Physics Revision Exam Physics Revision Examination Physics Revision Form One Physics Revision Notes Physics Revision Notes Physics Physics Revision Notes Form 1 Physics Revision Notes Form 2 Physics Revision Notes Form 3 Physics Revision Notes Form 4 Physics Revision Notes IGCSE Physics Revision Paper One Physics Revision Questions Physics Revision Questions and Answers Physics Revision Questions and Answers Form 1 Physics Revision Questions and Answers Form 2 Physics Revision Questions and Answers Form 3 Physics Revision Questions and Answers Form 4 Physics Revision Questions and Answers Form Four Physics Revision Questions and Answers Form One Physics Revision Questions and Answers Form Three Physics Revision Questions and Answers Form Two Physics Revision Questions Form 1 Physics Revision Questions Form 2 Physics Revision Questions Form 3 Physics Revision Questions Form 4 Physics Revision Questions Form Four Physics Revision Questions Form One Physics Revision Questions Form Three Physics Revision Questions Form Two Physics Revision Quiz Physics Revision Test Physics Secondary School Revision Physics Simple Notes Physics Spm Notes Download Physics Spm Notes Pdf Physics Spm Questions Physics Study Form 2 Physics Study Guide Physics Study Guide Answer Key Physics Study Guide Answers Physics Study Guide Physics Questions and Answers Physics Study Guide Ib Physics Study Guide Pdf Physics Study Guides Physics Study Notes Physics Study Notes Materials Form 1 Pdf Physics Study Notes Materials Form 2 3 Pdf Physics Study Notes Materials Form 2 Pdf Physics Study Notes Materials Form 3 Pdf Physics Study Notes Materials Form 4 Pdf Physics Syllabus in Kenya Physics Syllabus Pdf Physics Test 1 Quizlet Physics Test Questions Physics Test Questions and Answers Physics Test Questions and Answers Pdf Physics Topic One Form Four Physics Topics Form One Physics Unit 1 Quiz Physics Vol 3 Physics | Revision Physics Physics,form 4 Physics.form Four.topic Three PhysicsExam Form Three PhysicsModule Form 5 PhysicsNotes PhysicsNotes for Class 11 Pdf PhysicsNotes for Class 12 Pdf PhysicsNotes Form 1 PhysicsNotes Form 1 Free Download PhysicsNotes Form 2 PhysicsNotes Form 3 PhysicsNotes Form 3 Pdf PhysicsNotes IGCSE PhysicsNotes Pdf PhysicsPast Papers PhysicsQuestions and Answers Pdf PhysicsSimple Notes PhysicsSpm Notes Download PhysicsSpm Notes Pdf PhysicsSpm Questions PhysicsStudy Guide Answers PhysicsStudy Guide Pdf PhysicsStudy Guides Blologytextpapers Bridge Physics Business Past KCSE Past Papers Physics Form 3 Notes Pdf Physics Form 4 Notes Pdf C R E Form One KLB C R E Form One Oli Topic C.r.e Form 1 Notes Kenya C.r.e Form 2 Notes Kenya C.r.e Form 3 Notes C.r.e Form 3 Notes Kenya C.r.e Form 3 Pdf C.r.e Form 4 Notes Kenya C.r.e Form One Notes Pdf C.r.e Notes Form 1 C.r.e Revision Notes C.r.e Short Notes Cambridge IGCSE Physics Cambridge IGCSE Physics 3rd Edition Cambridge IGCSE Physics 3rd Edition Plus Cd South Asia Edition Cambridge IGCSE Physics Answers Cambridge IGCSE Physics Coursebook Pdf Download Cambridge IGCSE Physics Practical Workbook Cambridge IGCSE Physics Revision Guide Pdf Cambridge IGCSE Physics Study and Revision Guide 2nd Edition Pdf Cambridge IGCSE Physics Study and Revision Guide Pdf Cambridge IGCSE Physics Workbook Free Download Cambridge IGCSE Physics Workbook Pdf Cambridge IGCSE® Physics Coursebook Caucasian Chalk Circle Essay Questions Chapter 1 Introduction to Physics Chapter 1 Introduction to Physics Studies Cie a Level Physics Notes 2016 Cie a Level Physics Notes Pdf Cie Past Papers Class 10 Physics Chapter 1 Mcqs Class 8 Physics Notes KCSE-kcse College Physics Notes College Physics Practice Test College Physics Quiz College Physics Quiz Chapter 1 College Physics Quizlet College Physics Study Guide College Physics Study Guide Pdf College Physics Test Questions and Answers College Physics Volume 3 Pdf College PhysicsNotes Complete Physics for Cambridge IGCSE Complete Physics for Cambridge IGCSE Revision Guide Pdf County Mocks 2017 Cse Past Papers Physics 2017 Dl Physics Form 3 Pdf Kusoma Download Physics Form 1 Download Physics Form 2 Download Physics Form 2 Notes Download Physics Form 3 Download Physics Form 3 Notes Download Physics Form 4 Download Physics Form Four Download Physics Form One Download Physics Form Three Download Physics Form Two Download Physics Notes Form 3 Download Physics Notes Form One Download PhysicsNotes Form 3 Download Form Three Physics Notes Download Free KCSE Past Papers Physics Download Free KCSE Past Papers From KNEC. Download KCSE Past Papers With Answers Download KCSE Revision Notes Download KLB Physics Book 2 Download KLB Physics Book 3 Download KLB Physics Book 4 Download Notes of Physics Downloads | Physics | Form Four Exams | Exams Downloads | Physics | Form One Exams | Exams Downloads | Physics | Form Three Exams | Exams Downloads | Physics | Form Two Exams | Exams Downloads | KCSE Papers and Marking Schemes | Dvance KCSE Past Papers Easy Physics Questions Edexcel a Level Physics B Edexcel a Level Physics Notes Pdf Edexcel a Level Physics Salters Nuffield Edexcel A2 Physics Notes Edexcel as Physics Revision Guide Pdf Edexcel Physics A2 Revision Notes Pdf Edexcel Physics Unit 2 Revision Notes Edexcel GCSE Physics Revision Guide Pdf Edexcel IGCSE Physics Past Papers Edexcel IGCSE Physics Revision Guide Free Pdf Download Edexcel IGCSE Physics Revision Guide Pdf Edexcel IGCSE Physics Revision Guide Pdf Download Electronics Form Four Notes Energy Questions Physics Bowl Essay Questions and Answers KCSE Physics Notes Essay Questions and Answers on Betrayal in the City Essay Questions Based on Betrayal in the City Evolving World Physics Book 1 Pdf Evolving World Physics Book 4 Notes Evolving World Physics Book Form 1 Evolving World-history Book 3 Exam Notes for Physics 101 Exams KCSE Physics Paper 1 Questions and Answers F3 Physics Test Paper Find Download KCSE Past Papers With Answers Find KCSE Physics Essay Questions and Answers Form 1 Physics Exam Form 1 Physics Notes Form 1 Physics Questions and Answers Form 1 Physics Questions and Answers Pdf Form 1 Physics Revision Notes Form 1 Physics Summurized Revision Pdf Form 1 Physics Syllabus Form 1 Physics Test Paper Pdf Form 1 Physics Topics Form 1 PhysicsNotes Form 1 PhysicsQuestions and Answers Form 1 PhysicsRevision Notes Form 1 PhysicsSyllabus Form 1 PhysicsTest Paper Pdf Form 1 Past Papers Form 1 Past Papers With Answers Form 1 Revision Papers Form 1 Subjects in Kenya Form 2 Physics Exam Form 2 Physics Exam Paper Form 2 Physics Exam Paper 2016 Form 2 Physics Exam Paper Free Download Form 2 Physics Exam Paper With Answer Form 2 Physics Final Year Exam Paper 2 Form 2 Physics Notes Form 2 Physics Notes and Revision Questions Form 2 Physics Notes Pdf Form 2 Physics Past Papers Form 2 Physics Questions Form 2 Physics Questions and Answers Form 2 Physics Questions and Answers > Form 2 Physics Questions and Answers Pdf Form 2 Physics Revision Notes Form 2 Physics Short Notes Form 2 Physics Syllabus Form 2 PhysicsExam Paper Form 2 PhysicsExam Paper Free Download Form 2 PhysicsExam Paper With Answer Form 2 PhysicsFinal Year Exam Paper 2 Form 2 PhysicsPast Papers Form 2 PhysicsRevision Notes Form 2 PhysicsShort Notes Form 2 PhysicsSyllabus Form 2 Revision Papers Form 2 Subjects in Kenya Form 3 Physics Book Form 3 Physics Exam Form 3 Physics Exam Paper Form 3 Physics Notes Form 3 Physics Past Papers Form 3 Physics Questions Form 3 Physics Questions and Answers Form 3 Physics Questions and Answers Pdf Form 3 Physics Revision Notes Form 3 Physics Syllabus Form 3 PhysicsExam Paper Form 3 PhysicsNotes Form 3 PhysicsPast Papers Form 3 PhysicsQuestions Form 3 PhysicsQuestions and Answers Pdf Form 3 PhysicsRevision Notes Form 3 PhysicsSyllabus Form 3 C.r.e Form 3 Notes of Physics Topic on Fish Form 3 Past Papers Form 3 Revision Papers Form 3 Subjects in Kenya Form 4 Physics Exam Form 4 Physics Notes Form 4 Physics Notes Pdf Form 4 Physics Questions and Answers Form 4 Physics Questions and Answers Pdf Form 4 Physics Revision Notes Form 4 Physics Syllabus Form 4 Physics Topics Form 4 PhysicsNotes Form 4 PhysicsRevision Notes Form 4 PhysicsSyllabus Form 4 PhysicsTopics Form 4 Exam Papers Form 4 Revision Papers Form 4 Subjects in Kenya Form 5 Physics Topics Form 5 PhysicsTopics Form Five Physics Notes Form Five PhysicsNotes Form Four Physics Book Form Four Physics Notes Form Four Physics Notes Pdf Form Four Physics Questions and Answers Form Four Physics Questions and Answers Pdf Form Four Physics Revision Questions Form Four Physics Syllabus Form Four Physics Topics Form Four PhysicsNotes Form Four PhysicsQuestions and Answers Form Four PhysicsQuestions and Answers Pdf Form Four PhysicsTopics Form Four Notes Form Four Revision Papers Form Four Subjects in Kenya Form One Physics Book Form One Physics Examination Form One Physics First Topic Form One Physics Lesson Plan Form One Physics Notes Pdf Form One Physics Past Papers Pdf Form One Physics Questions Form One Physics Questions and Answers Form One Physics Questions and Answers Pdf Form One Physics Revision Questions Form One Physics Short Notes Form One Physics Syllabus Form One Physics Topics Form One PhysicsExamination Form One PhysicsPast Papers Pdf Form One PhysicsQuestions and Answers Form One PhysicsQuestions and Answers Pdf Form One PhysicsTopics Form One Exams Form One Notes of Physics Form One Past Papers Form One Subjects in Kenya Form One Term One Physics Exam Form One Term One PhysicsExam Form Three Physics Book Form Three Physics Book Pdf Form Three Physics Notes Form Three Physics Notes Pdf Form Three Physics Questions and Answers Form Three Physics Questions and Answers Pdf Form Three Physics Revision Questions Form Three Physics Syllabus Form Three Physics Topics Form Three PhysicsNotes Form Three PhysicsNotes Pdf Form Three PhysicsQuestions and Answers Form Three PhysicsQuestions and Answers Pdf Form Three PhysicsTopics Form Three Subjects in Kenya Form Two Physics Book Form Two Physics Cat Form Two Physics Examination Form Two Physics Notes Form Two Physics Notes Pdf Form Two Physics Past Papers Form Two Physics Questions and Answers Form Two Physics Questions and Answers Pdf Form Two Physics Revision Questions Form Two Physics Syllabus Form Two Physics Topics Form Two PhysicsNotes Form Two PhysicsNotes Pdf Form Two PhysicsQuestions and Answers Form Two PhysicsQuestions and Answers Pdf Form Two PhysicsSyllabus Form Two PhysicsTopics Form Two Notes Form Two Subjects in Kenya Free a-level Physics Revision App | Pass Your Physics Exams Free Physics Form 1 Notes Free Physics Notes Form 1 Free Physics Notes Pdf Free PhysicsNotes Pdf Free College Physics Practice Test Free Form1,form2,form3 Past Papers Free KCSE Past Papers Free KCSE Mocks 2015 Free KCSE Past Papers 2014 Free KCSE Past Papers KCSE Past Free KCSE Past Papers Kenya, Free KCSE Past Papers With Answers Free KCSE Questions and Answers on Physics Free KCSE Revision Notes Free Marking Schemes Free Mocks Online KCSE Answers Past Exams Question Papers Free Revision Papers From Three Notes Topic One KLB Fun Physics Questions Funny Physics Questions Funny Physics Questions and Answers Funny Physics Questions to Ask Funny Physics Quotes GCSE Physics Exam Questions and Answers GCSE Physics Past Papers GCSE Physics Revision GCSE Physics Revision Notes GCSE Physics Revision Notes Pdf GCSE Physics Revision Notes Pdf 9-1 GCSE Physics Revision Questions and Answers GCSE Physics Textbook Pdf GCSE Physics Topics Pass My Exams: Easy Exam Revision Notes Home Physics Notes Pdf Home Physics Practice Test With Answers Home Physics Quiz Home Physics Quiz Pdf Home Physics Test Questions and Answers Home Physics Test Questions and Answers Pdf Home Knowledge in Physics Human Body Good Physics Questions to Ask GRE Physics Practice Test GRE Physics Subject Test Pdf Handbook of Physics Pdf Free Download Hard Physics Questions Hard Physics Questions and Answers Hard Physics Questions to Ask Your Teacher Hard Physics Quiz Questions Hard Form 3 Physics Question High School Physics Final Exam Doc High School Physics Final Exam Pdf High School Physics Final Exam Questions High School Physics Final Exam Questions and Answers High School Physics Notes High School Physics Practice Test High School Physics Pretest With Answers High School Physics Questions and Answers Pdf High School Physics Study Guide High School Physics Test Questions and Answers Pdf High School PhysicsNotes High School PhysicsStudy Guide How to Answer KCSE Physics Question How to Motivate a Form 4 Student How to Motivate a KCSE Candidate How to Motivate a KCSE Student How to Pass Physics Questions & Answers Form 1&2 | Text Book How to Revise Physics How to Revise Effectively for KCSE How to Study Physics: 5 Study Techniques to Master Physics Hsc Physics 2018 Hsc Physics 2019 Https://www.knec.ac.ke/ Www.knec-portal.ac.ke/ KNEC Portal: Ial Physics Notes Ib Physics Cold War Notes Ib Physics Notes Ib Physics Notes Pdf Ib Physics of the Americas Notes Ib Physics of the Americas Study Guide Ib Physics Paper 2 Study Guide Ib Physics Question Bank by Topic Ib Physics Study Guide Pdf Ict Notes Form 1 IGCSE Physics Alternative to Practical Revision IGCSE Physics Alternative to Practical Revision Notes IGCSE Physics Book IGCSE Physics Book Pdf Download IGCSE Physics Notes IGCSE Physics Notes 2017 Pdf IGCSE Physics Notes Edexcel IGCSE Physics Paper 2 Notes IGCSE Physics Paper 6 Notes IGCSE Physics Past Papers IGCSE Physics Past Papers 2014 IGCSE Physics Past Papers 2017 IGCSE Physics Pdf IGCSE Physics Pre Release Material 2018 IGCSE Physics Resources IGCSE Physics Revision Guide IGCSE Physics Revision Guide Free Download IGCSE Physics Revision Guide Pdf Download IGCSE Physics Revision Notes Pdf IGCSE Physics Revision Worksheets IGCSE Physics Workbook Pdf IGCSE Physics Znotes IGCSE PhysicsPast Papers IGCSE Notes Physics Importance of Agroforestry Inorganic Physics Multiple Choice Questions With Answers Pdf Inorganic Physics Questions and Answers Pdf Interesting Physics Questions Interesting Physics Questions and Answers Interesting Questions to Ask About Physics Intro to Physics Quiz Introduction of Physics Form One Introduction to Physics Introduction to Physics Notes Introduction to Physics Pdf Introduction to PhysicsNotes Is Agroforestry Sustainable? K.c.s.e Answers Physics Paper One 2018 K.c.s.e Physics 2017 K.c.s.e Physics 2018 K.c.s.e Physics Paper 1 2017 K.c.s.e Mocks 2018 K.c.s.e Papers 2015 K.c.s.e Papers 2016 K.c.s.e Past Papers 2014 K.c.s.e.Physics Paper 2 Year 2018 K.c.s.e.results 2018 for Busia County K.l.b Physics Form 3 K.l.b Physics Notes K.l.b PhysicsNotes Kasneb Past Papers for Colleges Physics Past Papers KCSE 2010 Marking Scheme KCSE 2010 Past Papers KCSE 2011 Physics Paper 1 KCSE 2011 Marking Scheme KCSE 2012 Physics Paper 2 Marking Scheme KCSE 2012 Marking Schemes KCSE 2013 Physics Paper 1 KCSE 2013 Marking Scheme KCSE 2013 Marking Scheme Pdf KCSE 2014 KCSE 2015 Physics Paper 2 KCSE 2015 Physics Paper 3 KCSE 2015 Marking Scheme KCSE 2015 Past Papers KCSE 2016 Physics Paper 1 KCSE 2016 Physics Paper 2 KCSE 2017 Physics Paper 1 KCSE 2017 Physics Paper 2 KCSE 2017 Hostory Papers With Answers.com KCSE 2017 Marking Scheme KCSE 2017 Papers KCSE 2017 Papers and Marking Scheme KCSE 2017 Papers Pdf KCSE 2017 Past Papers KCSE 2017 Prediction Pdf KCSE 2018 Physics and Answers KCSE 2018 Physics Prediction KCSE 2018 Leakage KCSE 2018 Marking Scheme KCSE 2018 Papers KCSE 2018 Prediction Pdf KCSE 2018 Predictions KCSE 2018 Questions KCSE 2018 Questions and Answers KCSE 2019 Leakage Physics KCSE 2019 Marking Scheme KCSE 2019 Questions KCSE 2019 Questions and Answers KCSE 2020 Questions KCSE 2020 Questions and Answers KCSE Answers KCSE Answers Past Exams Question Papers Downloads | KCSE Physics 2011 KCSE Physics 2016 KCSE Physics Diagramsbiology Revision Tips KCSE Physics Essay Questions and Answers KCSE Physics Essay Questions and Answers Pdf KCSE Physics Essays KCSE Physics Essays Pdf KCSE Physics Marking Schemes KCSE Physics Notes KCSE Physics Notes Pdf KCSE Physics Notes, Syllabus, Questions, Answers KCSE Physics Paper 1 KCSE Physics Paper 1 2011 KCSE Physics Paper 1 2012 KCSE Physics Paper 1 2013 KCSE Physics Paper 1 2015 KCSE Physics Paper 1 2016 KCSE Physics Paper 1 2017 KCSE Physics Paper 1 2017 Pdf KCSE Physics Paper 1 Questions and Answers KCSE Physics Paper 2 KCSE Physics Paper 2 2012 KCSE Physics Paper 2 2012 KCSE Physics Paper 2 2015 KCSE Physics Paper 2 2013 KCSE Physics Paper 2 2014 KCSE Physics Paper 2 2015 KCSE Physics Paper 2 2016 KCSE Physics Paper 2 2017 KCSE Physics Paper 3 KCSE Physics Paper 3 2012 KCSE Physics Paper 3 2016 KCSE Physics Paper 3 2017 KCSE Physics Paper 3 Past Papers KCSE Physics Past Papers KCSE Physics Past Papers and Answers KCSE Physics Past Papers Pdf KCSE Physics Practical KCSE Physics Practical 2015 KCSE Physics Practical 2016 KCSE Physics Practical Past Papers KCSE Physics Practicals KCSE Physics Practicals KCSE Physics Paper 1 KCSE Physics Question and Answer KCSE Physics Questions and Answers KCSE Physics Questions and Answers Ap Physics KCSE Physics Revision KCSE Physics Revision Notes KCSE Physics Revision Papers KCSE Physics Revision Questions KCSE Physics Revision Questions and Answers KCSE Physics Syllabus KCSE PhysicsNotes KCSE PhysicsPaper 1 KCSE PhysicsPaper 2 KCSE PhysicsPaper 2 Pdf KCSE PhysicsSyllabus KCSE Business Paper 1 2016 KCSE Business Past Papers KCSE Physics Past Papers KCSE Essay Questions in Betrayal in the City KCSE Essays KCSE Exam Papers 2018 KCSE Exam Papers Answers KCSE Form 1 Physics Revision KCSE Form 2 Physics Revision KCSE Form 3 Physics Revision KCSE Form 4 Physics Revision KCSE Form Four Physics Revision KCSE Form One Physics Revision KCSE Form Three Physics Revision KCSE Form Two Physics Revision KCSE KCSE Past Papers KNEC KCSE Leakage KCSE Leakage Physics KCSE Made Familiar Physics KCSE Made Familiar Physics Pdf KCSE Marking Scheme 2016 KCSE Marking Schemes KCSE Marking Schemes 2017 KCSE Marking Schemes Pdf KCSE Mock Exams KCSE Mock Papers 2015 KCSE Mock Papers 2017 KCSE Mock Papers 2018 KCSE Mock Papers Pdf KCSE Mock Papers Pdf 2018 KCSE Mock Papers Pdf KCSE Past Papers KCSE Mocks 2017 KCSE Mocks 2018 KCSE Notes KCSE Online Notes KCSE Online Past Papers KCSE Online Registration KCSE Papers 2015 KCSE Papers and Marking Schemes | Exams KCSE Past Papers KCSE Past Papers 2007 KCSE Past Papers 2009 KCSE Past Papers 2010 KCSE Past Papers 2011 KCSE Past Papers 2011 Pdf KCSE Past Papers 2012 KCSE Past Papers 2013 KCSE Past Papers 2013knec KCSE Past Papers 2014 KCSE Past Papers 2014 Pdf KCSE Past Papers 2015 KCSE Past Papers 2015 Marking Schemes KCSE Past Papers 2015 Pdf KCSE Past Papers 2016 KCSE Past Papers 2016 Pdf KCSE Past Papers 2017 KCSE Past Papers 2017 Pdf KCSE Past Papers 2018 KCSE Past Papers Physics KCSE Past Papers Physics and Answers KCSE Past Papers Physics Pdf KCSE Past Papers Physics With Answers KCSE Past Papers Physicsand Answers KCSE Past Papers Physics and Answers KCSE Past Papers KCSE and Answers KCSE Past Papers KCSE and Answers Free Mocks Online KCSE Past Papers Marking Scheme KCSE Past Papers Pdf Download KCSE Past Papers Pdf Download KCSE 2013 KCSE Past Papers With Answers KCSE Past Papers Woodwork and Answers KCSE Prediction 2017 KCSE Prediction 2018 KCSE Prediction 2018 Pdf KCSE Prediction Papers 2018 KCSE Prediction Questions KCSE Prediction Questions 2018 KCSE Prediction Questions and Answers KCSE Questions KCSE Questions and Answers KCSE Questions and Answers. KCSE Questions on Physics KCSE Results, Online Registration, KCSE Result Slip. KCSE Revision KCSE Revision Notes KCSE Revision Notes Physics KCSE Revision Notes Pdf KCSE Revision Papers KCSE Revision Papers 2014 KCSE Revision Papers With Answers KCSE Revision Question for Physics KCSE Revision Questions KCSE Revision Questions and Answers KCSE Revision | Secondary School | Text Books | Text Book Centre KCSE Syllabus Pdf KCSE Trial 2017 KCSE Trial Exams 2017 Kenya Secondary School Physics Syllabus Kenya Secondary School Physics Syllabus Pdf Kenya Secondary School PhysicsSyllabus Pdf Kenya Secondary School Syllabus Pdf Kenya-kcse-christian Religious Education Syllabus Kenyaplex KCSE Past Papers Kenyaplex Past Papers for Secondary KLB Physics Book 1 Download KLB Physics Book 1 Notes KLB Physics Book 1 Pdf KLB Physics Book 2 KLB Physics Book 2 Notes KLB Physics Book 2 Notes Pdf KLB Physics Book 2 Pdf KLB Physics Book 3 Notes KLB Physics Book 3 Pdf KLB Physics Book 3 Pdf Download KLB Physics Book 4 Notes KLB Physics Book 4 Pdf KLB Physics Book 4 Pdf Download KLB Physics Book 4 Topics KLB Physics Book One KLB Physics Form 1 KLB Physics Form 1 Notes KLB Physics Form 1 Pdf KLB Physics Form 2 KLB Physics Form 2 Book KLB Physics Form 2 Notes KLB Physics Form 2 Pdf KLB Physics Form 2 Pdf Download KLB Physics Form 2 Schemes of Work KLB Physics Form 3 KLB Physics Form 3 Notes KLB Physics Form 3 Notes Pdf KLB Physics Form 3 Pdf KLB Physics Form 3 Pdf Download KLB Physics Form 4 KLB Physics Form 4 Notes KLB Physics Form 4 Pdf KLB Physics Form Four KLB Physics Form Four Notes KLB Physics Form One KLB Physics Form One Notes KLB Physics Form Three KLB Physics Form Three Notes KLB Physics Form Two KLB Physics Form Two Notes KLB Physics Notes KLB Physics Notes Form 4 KLB Physics Pdf KLB PhysicsNotes KLB PhysicsNotes Form 4 KLB PhysicsPdf KNEC Physics Syllabus KNEC Examiners Portal KNEC Website KNEC Ict Past Papers KNEC Past Papers for Colleges KNEC Past Papers Free Download KNEC Past Papers Free Downloads KNEC Past Papers Pdf KNEC Portal Confirmation KNEC Portal KCSE Results KNEC Portal KNEC Past Papers for Colleges Kasneb Past Papers KNEC Revision Papers KNEC Technical Exams Past Papers Kusoma Physics Notes Kusoma Physics Notes Pdf Kusoma Notes Physics Kusoma.co.ke Kusoma.com Past Papers Learner Guide for Cambridge IGCSE Physics Longhorn Physics Book 3 Pdf Made Familiar Physics Made Familiar Physics Pdf Made Familiar Physics Questions Maktaba Tetea Notes Marking Scheme KCSE Physics Past Papers Math Form2 Note Mcqs About Gaseous Exchange Middle School Physics Bowl Physics Questions Mock Past Papers 2017 Mock Past Papers With Answers Mokasa Mock 2017 More Than 1800 Physics Questions and Answers to Help You Study Multiple Choice Questions on Physics Necta Physics Past Papers Necta Physics Practicals Necta PhysicsPast Papers Necta PhysicsPracticals Necta Form Four Past Papers Necta Past Papers Form 4 Necta Past Papers Form 4 2016 Necta Past Papers Form Six Necta Past Papers Form Two Necta Questions and Answers Necta Review Questions Notes Physics Form 1 Notes Physics Form 2 Notes Physics Form 3 Notes Physics Form 3 Notes Pdf Notes Physics Form 3 Syllabus Notes Physics Form 4 Syllabus Notes on Physics Studies Notes Za Physics 4m 2 Notes Za Physics Form One Notes Za Physics Form Three O Level Physics Practical Experiments O Level Physics Questions and Answers Pdf Orm Three Physics Notes Page Navigation Papacambridge Physics IGCSE Papers KNEC KCSE Online Past Papers KNEC KCSE Results Past Papers Past KCSE Papers Past Paper Questions by Topic Physics Past Papers 2014 Past Papers in Kenya Pdf Physics Form 3 Pdf Physics Notes Pdf Physics Notes Form 1 Pdf Physics Notes Form 2 Pdf Physics Notes Form 3 Pdf Physics Notes Form 4 Pdf Physics Notes Form Four Pdf Physics Notes Form One Pdf Physics Notes Form Three Pdf Physics Notes Form Two Pdf Form 1 Physics Questions and Answers Pdf Form 2 Physics Questions and Answers Pdf Form 3 Physics Questions and Answers Pdf Form 4 Physics Questions and Answers Pdf Form Four Physics Questions and Answers Pdf Form One Physics Questions and Answers Pdf Form Three Physics Questions and Answers Pdf Form Two Physics Questions and Answers Pdf Free KCSE Past Papers and Marking Schemes Pdf" Revision Questions Physics Form 1 Practical Physics Experiments Pdf Practical Physics Question and Answer Pdf Pre Mocks 2018 Preliminary Physics Primary and Secondary Tillage Implements Ppt Pte KNEC Past Papers Questions and Answers Pdf Physics Form 1 Questions and Answers Pdf Physics Form 2 Questions and Answers Pdf Physics Form 3 Questions and Answers Pdf Physics Form 4 Questions and Answers Pdf Physics Form Four Questions and Answers Pdf Physics Form One Questions and Answers Pdf Physics Form Three Questions and Answers Pdf Physics Form Two Questions Based to Introduction to Physics Questions on Gaseous Exchange in Humans Questions on Introduction to Physics Questions to Ask in Physics Class Questions to Confuse Your Physics Teacher Quizlet Physics Test Quizlet Test Questions Qustions in Physics and Answers Revision Revision Physics Notes and Questions? Revision Quiz for Physics for Form Three S.1 Physics Questions S.2 Physics Questions S.3 Physics Questions S.4 Physics Questions Sample Essays on Betrayal in the City School Physics Notes Secondary Physics Notes Secondary Physics Notes Pdf Secondary PhysicsNotes Pdf Senior 1 Physics Notes Senior 2 Physics Notes Senior 3 Physics Notes Senior 4 Physics Notes Senior 5 Physics Notes Senior 6 Physics Notes Senior Five Physics Notes Senior Four Physics Notes Senior One Physics Notes Senior Six Physics Notes Senior Three Physics Notes Senior Two Physics Notes Simple Scientific Questions Smart Questions to Ask a Physics Teacher Snab Physics Revision Notes Southwest Mock Paper 2 2016 Physics Only Spm Physics Revision Notes Spm Notes Success Physics Spm Pdf Success PhysicsSpm Pdf Summary of Physics Form 3 Tahossa Past Papers To Motivate a Form 4 KCSE Student To Motivate a Form 4 Student Topical Revision Material Tricky Physics Questions and Answers Tricky Physics Questions for Adults Tricky Physics Questions With Answers Tricky Physics Quiz Questions Two Physics Revision Questions University Physics Volume 3 Openstax University Physics Volume 3 Pdf University Physics Volume 4 Pdf Ur Revision Guide IGCSE Physics What Are the Types of Gametes Working of Excretory System Www.Physics Form One Notes.com Www.Physics From One KLB.com Www.form 1 Physics.com Www.form 2 Physics.com Www.form 3 Physics.com Www.form 4 Physics.com Www.form Four Physics.com Www.form One Physics.com Www.form Three Physics.com Www.form Two Physics.com Www.kusoma Notes Www.kusoma Revision Materials Www.kusoma.co.ke Physics Notes Xtremepapers IGCSE Physics Year 11 Physics Z Notes Physics IGCSE Znotes as Physics Form 1 Summarized Notes For Phyics Form 2 Summarized Notes For Phyics Form 3 Summarized Notes For Phyics Form 4 Summarized Notes For Phyics Form 1 Summarized Notes For Physics Form 2 Summarized Notes For Physics Form 3 Summarized Notes For Physics Form 4 Summarized Notes For Physics 2018 KCSE PHYSICS PRACTICAL QUESTIONS Physics Paper One 2017 Physics Paper Two 2017 Physics Paper Three 2017 Physics Paper 1 2017 Physics Paper 2 2017 Physics Paper 3 2017 A Level Physics Questions and Answers Pdf All Physics Questions and Answers Pdf,ppt Physics Exam Questions and Answers Pdf Physics Multiple Choice Questions and Answers Pdf Physics Questions and Answers for High Schools Pdf Physics Notes Form 1-4 Pdf Free High School Notes Kenya Free Kcse Revision Notes General Physics Test Questions and Answers Pdf High School Physics Multiple Choice Questions and Answers Pdf Kcse Physics Revision Notes Pdf Kcse Revision Books Pdf Kcse Revision Notes Pdf Kenya Secondary School Notes Pdf Notes of Form 123 and 4 All Subject Physics Notes Form 1 Free Download Advice to KCSE Candidates Best Revision Books for KCSE How to Pass an Exam Successfully How to Pass KCSE 2018 How to Pass KCSE 2019 How to Pass KCSE Physics Paper How to Pass KCSE Physics How to Pass KCSE Physics How to Pass KCSE 2019 K.c.s.e Physics Paper 1 2017 KCSE 2019 Prediction KCSE Prediction 2019 KCSE Revision Tips KCSE Physics Paper 1 2018 KCSE Physics Paper 2 2018 KCSE Physics Past Papers Pdf KCSE Past Papers 2012 KCSE Past Papers 2017 Pdf KCSE Past Papers 2018 KCSE Past Papers of Physics Pp2 KCSE Physics Past Papers Pdf Physics KCSE Papers With Their Marking Schemes Physics Paper 1 and Answers KCSE 2017 Papers and Marking Scheme KCSE 2019 Papers and Marking Scheme KCSE Physics Paper 1 2018 KCSE Physics Paper 1 2019 KCSE Physics Paper 1 2019 Past Papers KCSE Physics Paper 2 2017 KCSE Physics Paper 2 2018 KCSE Physics Paper 2 2019 KCSE Physics Paper 2 2019 Past Papers KCSE Physics Paper 3 2019 Past Papers KCSE Physics Past Papers and Answers KCSE Marking Schemes Pdf KCSE Past Papers 2015 Marking Schemes KCSE Past Papers 2019 Marking Schemes KCSE Past Papers Physics Paper 1 2019 KCSE Past Papers Physics Paper 2 2019 KCSE Past Papers Physics Paper 3 2019 Past Papers KCSE Physics Paper 1 2019 Past Papers KCSE Physics Paper 2 2019 Past Papers KCSE Physics Paper 3 2019 4m1 Notes Viusasa 4m2 Notes Viusasa 4m3 Notes Viusasa 4m4 Notes Viusasa Download on Viusasa - Download Now for Free Elimu - Viusasa Elimu Library | Notes, Exams, Lesson Plans, Schemes Elimu Online High School Notes - Revision Materials for Kenyan Schools Https //www.viusasa.com/elimu Kenya Notes Viusasa Elimu Form 1 Notes Viusasa Elimu Form 2 Notes Viusasa Elimu Form 3 Notes Viusasa Elimu Form 4 Notes Viusasa Elimu Form Four Notes Viusasa Elimu Form One Notes Viusasa Elimu Form Three Notes Viusasa Elimu Form Two Viusasa Viusasa Education Viusasa Elimu Viusasa Elimu Class 6 Viusasa Elimu Form 1 Viusasa Elimu Form 1 Notes Viusasa Elimu Form 2 Viusasa Elimu Form 2 Notes Viusasa Elimu Form 3 Viusasa Elimu Form 3 Notes Viusasa Elimu Form 4 Viusasa Elimu Form 4 Notes Viusasa Elimu Form Four Viusasa Elimu Form Four Notes Viusasa Elimu Form One Viusasa Elimu Form One Notes Viusasa Elimu Form Three Viusasa Elimu Form Three Notes Viusasa Elimu Form Two Viusasa Elimu Form Two Notes Viusasa High School Notes - Revision Materials for Kenyan Schools Viusasa Notes Physics Mock Papers Physics Paper 1 2019 Physics Paper 1 Notes Physics Paper 1 Questions and Answers Physics Paper 1 Questions and Answers Pdf Physics Paper 2 Form 3 Physics Paper 2 Notes Physics Paper 2 Questions and Answers Pdf Physics Past Papers Physics Past Papers Pdf Physics Questions and Answers Pdf Physics Revision Questions and Answers Pdf Physics Revision Questions Pdf Common Exam Questions in Physics Paper 1 Common Exam Questions in Physics Paper 2 Common Test Questions in Physics Paper 1 Common Tested Questions in Physics Paper 1 Commonly Tested Questions in Physics Paper 1 K.c.s.e.Physics Questions and Answers KCSE 2019 Physics Paper 1 Marking Scheme KCSE 2019 Physics Paper 2 KCSE 2019 Physics Paper 2 Marking Scheme KCSE 2020 Prediction Questions and Answers KCSE Physics Paper 1 2019 KCSE Physics Paper 2 2016 KCSE Physics Paper 2 2017 KCSE Physics Paper 2 2018 KCSE Physics Paper 2 2019 KCSE Physics Questions and Answers Most Tested Questions in Physics Paper 1 Most Tested Questions in Physics Paper 2 Mostly Tested Questions in Physics Paper 1 Mostly Tested Questions in Physics Paper 2 A Level Physics Notes Uganda Pdf Download Buddo Junior School Holiday Work Physics Notes for a Level Pdf Physics Notes Form 1 Physics Notes Form 3 the Mole Physics Notes Form 4 Physics Notes O Level Physics Notes O Level Uganda Physics Notes O Level Uganda Pdf Physics Notes Pdf Download Physics Notes Form 3 Physics Notes Form 3 Physics Questions and Answers Form Three Physics Syllabus Pdf Gayaza High School Physics Notes Gayaza High School Physics Notes Pdf Gayaza High School Physics Past Papers Gayaza High School Physics Past Papers Answers Gayaza High School Physics Past Papers Questions and Answers Gayaza High School E Learning Gayaza High School Elearning Platform Gayaza High School Examinations Gayaza High School Holiday Work Gayaza High School Notes Gayaza High School Notes 2021 Gayaza High School Notes 2022 Gayaza High School Notes 2023 Gayaza High School Notes Pdf Gayaza Junior School E Learning Platform Gayaza Junior School Holiday Work Gayaza O Level Physics Notes Home › Gayaza High School | Elearning Platform Kabojja Junior School Holiday Work Pdf Klb Physics Book 3 Klb Physics Form 3 Teachers Guide Klb Physics Notes List of Ugandan E-learning Platforms for Students Mirembe Junior School Holiday Work Ntare School Past Papers O Level Physics Notes Free Download O Level Physics Notes in Uganda O Level Physics Notes Pdf O Level Physics Notes Uganda Pdf O Level Physics Notes Uganda Pdf Download O Level General - Home › Gayaza High School | Elearning S.1 Physics Notes | Standard High School Zzana S.3 Physics Notes S.4 Physics 2 Notes | Standard High School Zzana Seeta High School Elearning Seeta High School Holiday Work Seeta High School Notes Seeta High School Notes Pdf Seeta High School Past Papers Seeta High School Past Papers Pdf Senior 1 Physics Notes Senior 1 Physics Notes in Uganda Senior 1 Physics Notes Uganda Senior 1 Physics Questions Senior 1 Exams Senior 1 Work Senior 1 Work 2020 Senior 1 Work 2020 Uganda Senior 2 Physics Notes Senior 2 Physics Notes in Uganda Senior 2 Physics Notes Uganda Senior 2 Physics Questions Senior 2 Exams Senior 2 Work Senior 2 Work 2020 Senior 2 Work 2020 Uganda Senior 3 Physics Notes Senior 3 Physics Notes in Uganda Senior 3 Physics Notes Uganda Senior 3 Physics Questions Senior 3 Exams Senior 3 Work Senior 3 Work 2020 Senior 3 Work 2020 Uganda Senior 4 Physics Notes Senior 4 Physics Notes in Uganda Senior 4 Physics Notes Uganda Senior 4 Physics Questions Senior 4 Exams Senior 4 Work Senior 4 Work 2020 Senior 4 Work 2020 Uganda Senior Four Physics Notes Senior Four Physics Notes in Uganda Senior Four Physics Notes Uganda Senior Four Physics Questions Senior Four Exams Senior Four Work Senior Four Work 2020 Senior Four Work 2020 Uganda Senior One Physics Notes Senior One Physics Notes in Uganda Senior One Physics Notes Uganda Senior One Physics Questions Senior One Exams Senior One Work Senior One Work 2020 Senior One Work 2020 Uganda Senior Three Physics Notes Senior Three Physics Notes in Uganda Senior Three Physics Notes Uganda Senior Three Physics Questions Senior Three Exams Senior Three Work Senior Three Work 2020 Senior Three Work 2020 Uganda Senior Two Physics Notes Senior Two Physics Notes in Uganda Senior Two Physics Notes Pdf Senior Two Physics Notes Uganda Senior Two Physics Questions Senior Two Exams Senior Two Work Senior Two Work 2020 Senior Two Work 2020 Uganda St Mary's Kitende E Learning Standard High School Notes Standard High School Zana a Level Notes Standard High School Zana Com Notes Standard High School Zana E Learning Standard High School Zana E Learning Platform Standard High School Zana E-learning Platform Standard High School Zana Notes for Senior Two Standard High School Zana Notes Pdf Standard High School Zana Website Standard High Zana Notes Uace Physics Notes Uace Physics Notes Pdf Uce Physics Notes Pdf Uce Physics Notes Pdf Download Uce Past Papers Uganda Secondary Schools E-learning Platform Uneb Marking Guides Pdf Uneb Past Papers and Answers Pdf Physics Form 1 Questions and Answers Pdf Download Physics Notes Form 1-4 Physics Form 1 Questions and Answers Pdf Form 1 Physics Topical Questions Form 1 Physics Exam Paper With Answer Pdf Form 1 Exams 2020 Form 1 Exams 2021 Form 1 Exams 2022 Form 1 Exams 2023 Form 1 Physics Past Papers Form 1 Physics Revision Papers With Answers Sample Physics Test for Form One Exams Ntare School Past Papers Seeta High School Notes Pdf Seeta High School Past Papers Seeta High School Past Papers Pdf St Mary's Kitende Past Papers Uce Physics Notes Pdf Uce Past Papers Uneb Marking Guides Pdf Uneb Past Papers and Answers Pdf Physics Book 3 Download Physics Form 3 Questions and Answers Physics Form 3 Syllabus Physics Form 3 Topics Physics Notes Form 3 Physics Notes Form 3 Physics Notes Form 3 Physics Notes Form Three Physics Notes Pdf Download Form 3 Physics Exam Form 3 Physics Notes on Pdf Revision Physics Revision a Level Physics Revision Notes - Physics S.1-Physics-notes S.1-Physics-notes Uganda S.2-Physics-notes S.2-Physics-notes Uganda S.3-Physics-notes S.3-Physics-notes Uganda S.4-Physics-notes S.4-Physics-notes Uganda S.5-Physics-notes S.5-Physics-notes Uganda S.6-Physics-notes S.6-Physics-notes Uganda S1 Physics Notes Term 1 S1 Physics Notes Term 2 S1 Physics Notes Term 3 S2 Physics Notes Term 1 S2 Physics Notes Term 2 S2 Physics Notes Term 3 S3 Physics Notes Term 1 S3 Physics Notes Term 2 S3 Physics Notes Term 3 S4 Physics Notes Term 1 S4 Physics Notes Term 2 S4 Physics Notes Term 3 Senior 3 Physics Notes Senior 3 Physics Notes Uganda Senior Three Physics Notes Senior Three Physics Notes Uganda Physics Form 1 the Cell Physics Form One Notes Free – Education News Physics Notes Form 1-4 Physics Questions and Answers Form 1 - Physics Form One Physics Questions and Answers Form 2 - Physics Form Two Physics Questions and Answers Form 3 - Physics Form Three Physics Questions and Answers Form 4 - Physics Form Four Physics Questions and Answers Senior 1 - Physics Senior One Physics Questions and Answers Senior 2 - Physics Senior Two Physics Questions and Answers Senior 3 - Physics Senior Three Physics Questions and Answers Senior 4 - Physics Senior Four Physics Topic by Topic Questions and Answers Combined Science Notes Form 1 Form 1 Physics Revision Questions and Answers Form 1 Physics Topical Questions Form 1 Revision Papers With Answers Form 2 Physics Revision Questions and Answers Form 3 Physics Revision Questions and Answers Form Four Physics Revision Questions and Answers Form One Physics Revision Questions and Answers Form Three Physics Revision Questions and Answers Form Two Physics Revision Questions and Answers Free Physics Form 1 Notes Free Physics Notes Introduction to Physics Form One Kcse Past Papers: Physics Form 1 Kcse Past Papers: Physics Form 1 Topical Questions Kcse Past Papers: Physics Form 1 Topical Questions and Answers Most Tested Areas in Physics Kcse Most Tested Areas in Physics Kcse Exams Most Tested Areas in Physics Uneb Most Tested Areas in Form 1 in Physics Most Tested Areas in Form 2 in Physics Most Tested Areas in Form 3 in Physics Most Tested Areas in Form 4 in Physics Most Tested Areas in Form Four in Physics Most Tested Areas in Form One in Physics Most Tested Areas in Form Three in Physics Most Tested Areas in Form Two in Physics Most Tested Areas in Kcse Physics Most Tested Areas in Kcse Physics Exams Most Tested Areas in Senior 1 in Physics Most Tested Areas in Senior 2 in Physics Most Tested Areas in Senior 3 in Physics Most Tested Areas in Senior 4 in Physics Most Tested Areas in Senior Four in Physics Most Tested Areas in Senior One in Physics Most Tested Areas in Senior Three in Physics Most Tested Areas in Senior Two in Physics Most Tested Areas in Uneb Physics Revision Notes Physics Form 1 - Free Kcse Past Papers Who Was Physics Champion KCSE 2019 Top 100 Students in Physics KCSE 2019 Best 100 Students in Physics KCSE 2019 Top Student in Physics KCSE Best Student in Physics KCSE Who Was Physics Champion KCSE 2020 Top 100 Students in Physics KCSE 2020 Best 100 Students in Physics KCSE 2020 Names of Best Physics Students KCSE Names of Top Physics Students KCSE High School Physics Notes Pdf Klb Physics Form 1 Book Pdf Physics Form 1 Best Notes Physics Form 1 Notes Pdf Physics Form 1 Pressure Physics Form 1 Questions and Answers Physics Form 1 Questions and Answers Pdf Download Physics Form 2 Best Notes Physics Form 3 Best Notes Physics Form 4 Best Notes Physics Form Four Best Notes Physics Form One Best Notes Physics Form Three Best Notes Physics Form Two Best Notes Physics Notes Form 1 to 4 Physics Notes Pdf Klb Physics Form 1 Book Pdf Physics Form 1 Notes Pdf Physics Form 1 Pressure Physics Form 1 Questions and Answers Physics Form 1 Questions and Answers Pdf Download Physics Notes Form 1-4 Pdf Physics Notes Pdf Physics Summary Notes Pdf
Scholarship 2026/27
Current Scholarships 2026/2027 - Fully Funded
Full Undergraduate Scholarships 2026 - 2027
Fully Funded Masters Scholarships 2026 - 27
PhD Scholarships for International Students - Fully Funded!
Funding Opportunities for Journalists 2026/2027
Funding for Entrepreneurs 2026/2027
***
